Оксид азота и митохондриальное повреждение в ткани почек при моделировании искусственного кровообращения и циркуляторного ареста: экспериментальное исследование
Опубликован 15.04.2024
Ключевые слова
- искусственное кровообращение,
- митохондриальное повреждение,
- оксид азота,
- органопротекция,
- почка
- циркуляторный арест ...Показать
Как цитировать
Copyright (c) 2024 Бойко А.М., Каменщиков Н.О., Мирошниченко А.Г., Подоксенов Ю.К., Свирко Ю.С., Луговский В.А., Дьякова М.Л., Кравченко И.В., Мухомедзянов А.В., Маслов Л.Н., Базарбекова Б.А., Петлин К.А., Панфилов Д.С., Козлов Б.Н.

Это произведение доступно по лицензии Creative Commons «Attribution» («Атрибуция») 4.0 Всемирная.
Аннотация
Актуальность. Оперативные вмешательства в условиях искусственного кровообращения и циркуляторного ареста осложняются нарушением микроциркуляции в тканях, при этом ведущим патогенетическим фактором ишемически-реперфузионного повреждения является митохондриальная дисфункция при гипоксии. В этой связи поиск методов органопротекции крайне актуален, а применение оксида азота ввиду его плюрипотентных свойств выглядит многообещающим.
Цель. Изучить митохондриальное повреждение в биоптатах почек под воздействием искусственного кровообращения и циркуляторного ареста при моделировании кардиохирургической операции и оценить органопротекцию оксидом азота в эксперименте.
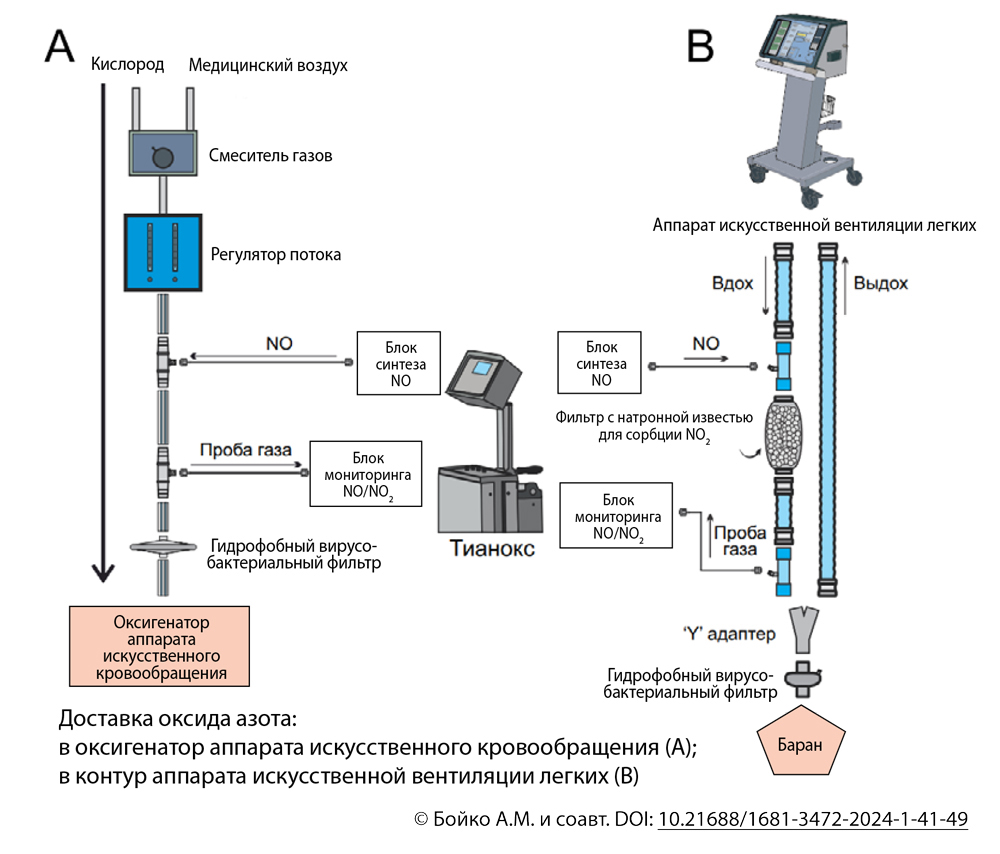
Методы. Исследование выполнено на баранах алтайской породы и включало моделирование искусственного кровообращения и циркуляторного ареста. В первой группе (n = 6) проводили интраоперационную донацию оксида азота. Во второй группе (n = 6) не осуществляли донацию оксида азота. Оценивали митохондриальное повреждение в биоптатах почек посредством измерения трансмембранного потенциала и Са2+-связывающей способности митохондрий, а также концентрации аденозинтрифосфата и лактата.
Результаты. Терапия оксидом азота сопровождалась меньшей степенью митохондриальной дисфункции в биоптатах почек по сравнению с контрольной группой.
Заключение. Энергетическое обеспечение тканей в условиях искусственного кровообращения и циркуляторного ареста в эксперименте улучшается на фоне донации оксида азота в концентрации 80 ppm.
Поступила в редакцию 11 февраля 2024 г. Исправлена 28 марта 2024 г. Принята к печати 29 марта 2024 г.
Финансирование
Исследование выполнено в рамках государственного задания «Защита органов оксидом азота в сердечно-сосудистой хирургии: технологическая поддержка (устройства синтеза и доставки), механизмы реализации защитных эффектов и влияние на клинические исходы» (тема № 122123000017-3).
Конфликт интересов
Авторы заявляют об отсутствии конфликта интересов.
Вклад авторов
Концепция и дизайн работы: А.М. Бойко, Н.О. Каменщиков, Ю.К. Подоксенов, Л.Н. Маслов, Б.Н. Козлов
Сбор и анализ данных: А.М. Бойко, Ю.С. Свирко,
В.А. Луговский, А.В. Мухомедзянов, Б.А. Базарбекова
Статистическая обработка данных: И.В. Кравченко
Написание статьи: А.М. Бойко
Исправление статьи: Н.О. Каменщиков, А.Г. Мирошниченко, Ю.К. Подоксенов, М.Л. Дьякова, К.А. Петлин, Д.С. Панфилов, Б.Н. Козлов
Утверждение окончательного варианта статьи: все авторы
Библиографические ссылки
- Каменщиков Н.О., Подоксенов Ю.К., Дьякова М.Л., Бойко А.М., Козлов Б.Н. Острое повреждение почек в кардиохирургии: определение, эпидемиология, исходы и социально-экономическая значимость. Патология кровообращения и кардиохирургия. 2020;24(4):11-21. https://doi.org/10.21688/1681-3472-2020-4-11-21 Kamenshchikov N.O., Podoksenov Y.K., Diakova M.L., Boyko A.M., Kozlov B.N. Acute kidney injury in cardiac surgery: definition, epidemiology, outcomes and socio-economic significance. Patologiya krovoobrashcheniya i kardiokhirurgiya = Circulation Pathology and Cardiac Surgery. 2020;24(4):11-21. (In Russ.) https://doi.org/10.21688/1681-3472-2020-4-11-21
- Scherner M., Weber C., Schmidt H., Kuhr K., Hamacher S., Sabashnikov A., Eghbalzadeh K., Mader N., Wahlers T., Wippermann J. Impact of urgent coronary artery bypass grafting on acute kidney injury: a matched cohort study. Med Klin Intensivmed Notfmed. 2022;117(2):152-158. PMID: 33471151. https://doi.org/10.1007/s00063-020-00769-x
- Harky A., Joshi M., Gupta S., Teoh W.Y., Gatta F., Snosi M. Acute kidney injury associated with cardiac surgery: a comprehensive literature review. Braz J Cardiovasc Surg. 2020;35(2):211-224. PMID: 32369303; PMCID: PMC7199993. https://doi.org/10.21470/1678-9741-2019-0122
- Amano K., Takami Y., Ishikawa H., Ishida M., Tochii M., Akita K., Sakurai Y., Noda M., Takagi Y. Lower body ischaemic time is a risk factor for acute kidney injury after surgery for type A acute aortic dissection. Interact Cardiovasc Thorac Surg. 2020;30(1):107-112. PMID: 31501854. https://doi.org/10.1093/icvts/ivz220
- Ghincea C.V., Reece T.B., Eldeiry M., Roda G.F., Bronsert M.R., Jarrett M.J., Pal J.D., Cleveland J.C. Jr., Fullerton D.A., Aftab M. Predictors of acute kidney injury following aortic arch surgery. J Surg Res. 2019;242:40-46. PMID: 31063910. https://doi.org/10.1016/j.jss.2019.03.055
- Lei C., Berra L., Rezoagli E., Yu B., Dong H., Yu S., Hou L., Chen M., Chen W., Wang H., Zheng Q., Shen J., Jin Z., Chen T., Zhao R., Christie E., Sabbisetti V.S., Nordio F., Bonventre J.V., Xiong L., Zapol W.M. Nitric oxide decreases acute kidney injury and stage 3 chronic kidney disease after cardiac surgery. Am J Respir Crit Care Med. 2018;198(10):1279-1287. PMID: 29932345; PMCID: PMC6290943. https://doi.org/10.1164/rccm.201710-2150OC
- Kamenshchikov N.O., Duong N., Berra L. Nitric oxide in cardiac surgery: a review article. Biomedicines. 2023;11(4):1085. PMID: 37189703; PMCID: PMC10135597. https://doi.org/10.3390/biomedicines11041085
- Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120(4):c179-c184. PMID: 22890468. https://doi.org/10.1159/000339789
- Kork F., Balzer F., Spies C.D., Wernecke K.D., Ginde A.A., Jankowski J., Eltzschig H.K. Minor postoperative increases of creatinine are associated with higher mortality and longer hospital length of stay in surgical patients. Anesthesiology. 2015;123(6):1301-1311. PMID: 26492475; PMCID: PMC4679549. https://doi.org/10.1097/ALN.0000000000000891
- Haase M., Devarajan P., Haase-Fielitz A., Bellomo R., Cruz D.N., Wagener G., Krawczeski C.D., Koyner J.L., Murray P., Zappitelli M., Goldstein S.L., Makris K., Ronco C., Martensson J., Martling C.R., Venge P., Siew E., Ware L.B., Ikizler T.A., Mertens P.R. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol. 2011;57(17):1752-1761. PMID: 21511111; PMCID: PMC4866647. https://doi.org/10.1016/j.jacc.2010.11.051
- Schurle A., Koyner J.L. CSA-AKI: incidence, epidemiology, clinical outcomes, and economic impact. J Clin Med. 2021;10(24):5746. PMID: 34945041; PMCID: PMC8706363. https://doi.org/10.3390/jcm10245746
- Brown J.K., Shaw A.D., Mythen M.G., Guzzi L., Reddy V.S., Crisafi C., Engelman D.T.; PeriOperative Quality Initiative and the Enhanced Recovery After Surgery Cardiac Workgroup. Adult cardiac surgery-associated acute kidney injury: joint consensus report. J Cardiothorac Vasc Anesth. 2023;37(9):1579-1590. PMID: 37355415. https://doi.org/10.1053/j.jvca.2023.05.032
- Hofmann F. A concise discussion of the regulatory role of cGMP kinase I in cardiac physiology and pathology. Basic Res Cardiol. 2018;113(4):1-11. PMID: 29934662. https://doi.org/10.1007/s00395-018-0690-1
- Lukowski R., Cruz Santos M., Kuret A., Ruth P. cGMP and mitochondrial K+ channels—Compartmentalized but closely connected in cardioprotection. Br J Pharmacol. 2022;179(11):2344-2360. PMID: 33991427. https://doi.org/10.1111/bph.15536
- Prokudina E.S., Naryzhnaya N.V., Mukhomedzyanov A.V., Gorbunov A.S., Zhang Y., Yaggi A.S., Tsibulnikov S.Y., Nesterov E.A., Lishmanov Y.B., Suleiman M.S., Oeltgen P.R., Maslov L.N. Effect of chronic continuous normobaric hypoxia on functional state of cardiac mitochondria and tolerance of isolated rat heart to ischemia and reperfusion: role of µ and delta2 opioid receptors. Physiol Res. 2019;68(6):909-920. PMID: 31647288. https://doi.org/10.33549/physiolres.933945
- Бойко А.М., Каменщиков Н.О., Мирошниченко А.Г., Подоксенов Ю.К., Серебрякова О.Н., Дзюман А.Н., Свирко Ю.С., Дымбрылова О.Н., Луговский В.А., Дьякова М.Л., Панфилов Д.С., Козлов Б.Н. Влияние доставки оксида азота на повреждение почек при моделировании искусственного кровообращения с циркуляторным арестом в эксперименте. Фундаментальная и клиническая медицина. 2023;8(3):18-25. https://doi.org/10.23946/2500-0764-2023-8-3-18-25 Boyko A.M., Kamenshchikov N.O., Miroshnichenko A.G., Podoksenov Yu.K., Serebryakova O.N., Dzyuman A.N., Svirko Yu.S., Dymbrylova O.N., Lugovskiy V.A., Diakova M.L., Panfilov D.S., Kozlov B.N. Influence of nitric oxide delivery on kidney damage in experimental model of cardiopulmonary bypass with circulatory arrest. Fundamental and Clinical Medicine. 2023;8(3):18-25. (In Russ.) https://doi.org/10.23946/2500-0764-2023-8-3-18-25
- Nagasaka Y., Fernandez B.O., Steinbicker A.U., Spagnolli E., Malhotra R., Bloch D.B., Bloch K.D., Zapol W.M., Feelisch M. Pharmacological preconditioning with inhaled nitric oxide (NO): organ-specific differences in the lifetime of blood and tissue NO metabolites. Nitric Oxide. 2018;80:52-60. PMID: 30114529; PMCID: PMC6198794. https://doi.org/10.1016/j.niox.2018.08.006
- Signori D., Magliocca A., Hayashida K., Graw J.A., Malhotra R., Bellani G., Berra L., Rezoagli E. Inhaled nitric oxide: role in the pathophysiology of cardio-cerebrovascular and respiratory diseases. Intensive Care Med Exp. 2022;10(1):28. PMID: 35754072; PMCID: PMC9234017. https://doi.org/10.1186/s40635-022-00455-6
- Poderoso J.J., Helfenberger K., Poderoso C. The effect of nitric oxide on mitochondrial respiration. Nitric Oxide. 2019;88:61-72. PMID: 30999001. https://doi.org/10.1016/j.niox.2019.04.005
- Shiva S., Sack M.N., Greer J.J., Duranski M., Ringwood L.A., Burwell L., Wang X., MacArthur P.H., Shoja A., Raghavachari N., Calvert J.W., Brookes P.S., Lefer D.J., Gladwin M.T. Nitrite augments tolerance to ischemia/reperfusion injury via the modulation of mitochondrial electron transfer. J Exp Med. 2007;204(9):2089-102. PMID: 17682069; PMCID: PMC2118713. https://doi.org/10.1084/jem.20070198
- Тё М.А., Каменщиков Н.О., Подоксенов Ю.К., Мухомедзянов А.В., Маслов Л.Н., Козлов Б.Н. Влияние доставки оксида азота на энергетическое обеспечение почечной ткани при проведении искусственного кровообращения: экспериментальное исследование. Сибирский журнал клинической и экспериментальной медицины. 2024;39(1):163-170. https://doi.org/10.29001/2073-8552-2022-592 Tyo M.A., Kamenshchikov N.O., Podoksenov Yu.K., Mukhomedzyanov A.V., Maslov L.N., Kozlov B.N. Effect of nitric oxide delivery on energy supply of renal tissue in cardiopulmonary bypass: an experimental study. The Siberian Journal of Clinical and Experimental Medicine. 2024;39(1):163-170. (In Russ.) https://doi.org/10.29001/2073-8552-2022-592
- Тё М.А., Каменщиков Н.О., Подоксенов Ю.К., Мухомедзянов А.В., Маслов Л.Н., Козлов Б.Н. Влияние донации оксида азота на выраженность митохондриальной дисфункции почечной ткани при моделировании искусственного кровообращения: экспериментальное исследование. Вестник интенсивной терапии им. А.И. Салтанова. 2023;(4):176-184. https://doi.org/10.21320/1818-474X-2023-4-176-184 Tyo M.A., Kamenshchikov N.O., Podoksenov Y.K., Mukhomedzyanov A.V., Maslov L.N., Kozlov B.N. The effect of nitric oxide donation on the severity of mitochondrial disfunction to the renal tissue in cardiopulmonary bypass simulation: an experimental study. Annals of Critical Care. 2023;(4):176-184. (In Russ.) https://doi.org/10.21320/1818-474X-2023-4-176-184
- Kumar A., Noda K., Philips B., Velayutham M., Stolz D.B., Gladwin M.T., Shiva S., D'Cunha J. Nitrite attenuates mitochondrial impairment and vascular permeability induced by ischemia-reperfusion injury in the lung. Am J Physiol Lung Cell Mol Physiol. 2020;318(4):L580-L591. PMID: 32073901; PMCID: PMC7191477. https://doi.org/10.1152/ajplung.00367.2018
- Jiang M., Bai M., Lei J., Xie Y., Xu S., Jia Z., Zhang A. Mitochondrial dysfunction and the AKI-to-CKD transition. Am J Physiol Renal Physiol. 2020;319(6):F1105-F1116. PMID: 33073587. https://doi.org/10.1152/ajprenal.00285.2020
- Фрелих Г.А., Поломеева Н.Ю., Васильев А.С., Удут В.В. Современные методы оценки функционального состояния митохондрий. Сибирский журнал клинической и экспериментальной медицины. 2013;28(3):7-13. Frelikh G.A., Polomeeva N.U., Vasilev A.S., Udut V.V. State-of-the art methods of evaluation of mitochondrial function. The Siberian Journal of Clinical and Experimental Medicine. 2013;28(3):7-13. (In Russ.)

