Automated selection of signal frequency filtering parameters for monitoring sensory evoked potentials: a pilot study
Published 2025-04-28
Keywords
- Band-Pass Filter,
- Evoked Potentials,
- Intraoperative Neurophysiological Monitoring,
- Personalized Medicine,
- Signal-to-Noise Ratio
How to Cite
Copyright (c) 2025 Levin E.A., Pisarev Ya.V., Mukhina I.G., Glushaeva A.A., Kilchukov M.G.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Introduction: The monitoring of somatosensory, visual and auditory evoked potentials (EP) is used to control the preservation of the corresponding functions. To detect promptly signs of their impairment, it is necessary to minimize the time intervals between consecutive recordings of EP. However, the low amplitude of the latter forces researchers to resort to averaging at recording them that involves the long-term accumulation of tens or hundreds of responses to repeated stimuli. Frequency filtering improves the signal-to-noise ratio and reduces the number of required averagings, but its optimal parameters vary among patients and their selection requires considerable time and highly qualified personnel.
Objective: The study aimed to develop and test a method for automated selection of signal frequency filtering parameters using real data from intraoperative monitoring (IOM) of somatosensory and visual EPs and compare the results of automated selection with the choice made by an experienced neurophysiologist during surgery.
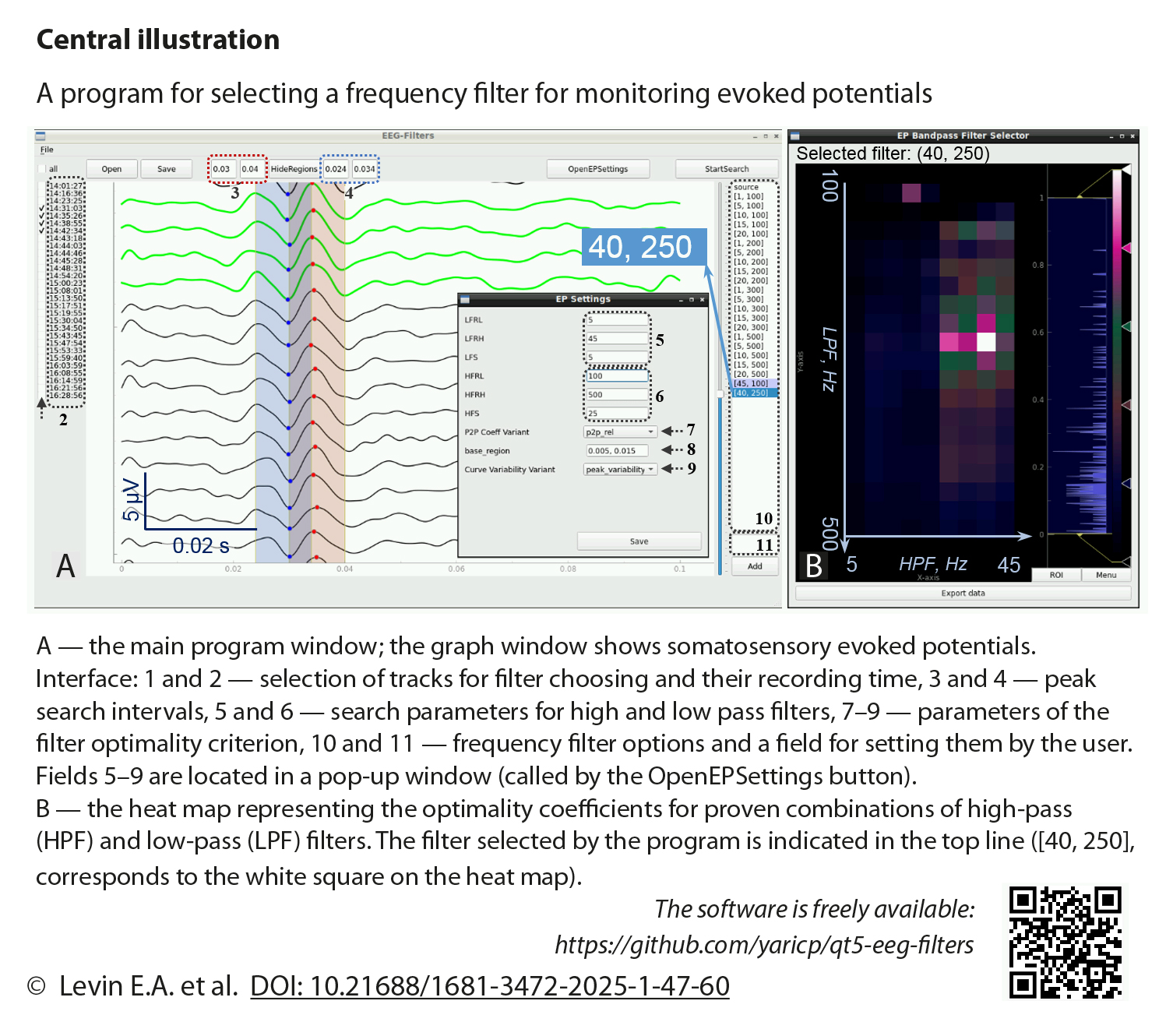
Methods: The automated frequency filter selection technique was implemented in our own program, qt5-eeg-filters. It loads unfiltered EPs, filters them using a user-defined set of filters, compares the filtering results taking into account the reproducibility and amplitudes of the EPs, and provides the user with the recommended passband of the frequency filter. Unfiltered EPs of 23 patients (79 records) who underwent intraoperative neuromonitoring with personalized selection of filtering parameters were automatically analyzed using four variants of the filter optimality criterion. These variants were obtained by combining two parameters: (1) the method of assessing the reproducibility of EPs only by characteristics of the peaks (PV) or by a curve segment (CV) and (2) the peak amplitude values used, namely absolute (Abs) or relative values compared to background oscillations (Rel). For each variant, the correlation coefficients of the filtering parameters proposed by the program with those actually selected during IOM were calculated, and their differences were assessed. In addition, the proposed filters were evaluated by a neurophysiologist on a five-point scale for subsequent comparison of the four variants of the optimality criterion with each other. Nonparametric methods were used for statistical analysis: Spearman correlation and Friedman/Wilcoxon test.
Results: With the optimality criteria of the AbsCV, RelCV, and RelPV types, the lower limit of the passband was higher than that selected by the neurophysiologist during IOM: p=0.00002, p=0.00003, and p=0.056, respectively, for visual EP (VEP), and p<0.00001, p<0.00001, and p=0.00002, respectively, for somatosensory EP (SSEP). With the same criteria, the upper limit of the passband was lower than that selected during IOM: p=0.00029, p=0.00002, and p=0.00037, respectively, for VEP, and p<0.00001, p<0.00001, and p=0.0002, respectively, for SSEP. Thus, with all these criteria, the program suggested using more "aggressive" filtering than the neurophysiologist actually used during the IOM (hereinafter referred to as "IOM filter"). With the optimality criterion of the AbsPV type, no clear trend was revealed; at the same time, the quality assessments of both SSEP and VEP selected with this criterion were the worst. The best quality assessments of the filters were obtained using the RelCV criterion for VEP (they did not differ significantly from the assessments for the IOM filter, p = 0.22) and AbsCV for SSEP (they were, however, worse than for the IOM filter, p = 0.0025).
For both VEP and SSEP, positive correlations were observed between the quality assessments when using the IOM filter and the filters proposed by the program. For VEP they were 0.74 (p=0.00014), 0.72 (p=0.0003), 0.74 (p=0.00016) and 0.56 (p=0.019) when comparing the IOM filter with the filters obtained using the RelCV, AbsCV, RelPV and AbsPV criteria, respectively. For SSEP the corresponding correlations were 0.23 (p=0.085), 0.49 (p=0.00014), 0.45 (p=0.00050) and 0.57 (p=0.00001).
In all cases when there were events that impaired the monitored function during IOM, the changes in EPs were reliably identified using both the IOM filters and the filters proposed by the program. However, the number of such events was insufficient for statistical analysis.
Conclusion: We demonstrated the possibility of automated selection of frequency filtering parameters for sensory evoked potentials using a dataset obtained during real intraoperative neuromonitoring sessions. Prospects for the development of the method are associated with the extension of the analysis to the single-trial level.
Received 2 December 2024. Accepted 11 December 2024.
Funding
The study was supported by the Russian Science Foundation, grant No. 23-25-00322. The Foundation did not influence the development of the concept and design of the study, data collection and processing, interpretation of the results, preparation, publication, and other stages of the article creation.
Conflict of interest
The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: E.A. Levin, Ya.V. Pisarev
Data collection and analysis: I.G. Mukhina, M.G. Kilchukov, A.A. Glushaeva, E.A. Levin
Statistical analysis: M.G. Kilchukov, A.A. Glushaeva, E.A. Levin
Drafting the article: E.A. Levin, Ya.V. Pisarev
Critical revision of the article: E.A. Levin, I.G. Mukhina
Final approval of the version to be published: E.A. Levin, Ya.V. Pisarev, I.G. Mukhina, A.A. Glushaeva, M.G. Kilchukov
References
- Дмитриев А.Ю., Синкин М.В., Дашьян В.Г. Интраоперационный нейрофизиологический мониторинг в хирургии опухолей головного мозга супратенториальной локализации. Часть 2. Исследование сенсорной проводимости, влияние на исходы и ограничения метода. Нейрохирургия. 2022;24(3):73-79. https://doi.org/10.17650/1683-3295-2022-24-3-73-79 Dmitriev A.Yu., Sinkin M.V., Dashyan V.G. Intraoperative neuromonitoring in surgery of supratentorial brain tumors. Part 2. Assessment of sensory conductivity, impact at outcomes and method restrictions. Neyrokhirurgiya = Russian Journal of Neurosurgery. 2022;24(3):73-79. (In Russ.) https://doi.org/10.17650/1683-3295-2022-24-3-73-79
- Sloan T.B., Edmonds H.L. Jr., Koht A. Intraoperative electrophysiologic monitoring in aortic surgery. J Cardiothorac Vasc Anesth. 2013;27(6):1364-1373. PMID: 23276593. https://doi.org/10.1053/j.jvca.2012.09.027
- Бобряков Н.А., Петров С.И., Середа Э.В., Москалев А.Г., Пономарев А.А., Казанков И.Ю., Седова Е.Ю., Максимова Е.Н. Интраоперационный нейрофизиологический мониторинг у пациентов с отсроченной церебральной ишемией после клипирования разорвавшихся артериальных аневризм. Нейрохирургия. 2024;26(2):26-36. https://doi.org/10.17650/1683-3295-2024-26-2-26-36 Bobriakov N.А., Petrov S.I., Sereda E.V., Moskalev A.G., Ponomarev A.A., Kazankov I.Yu., Sedova E.Yu., Maksimova E.N. Intraoperative neurophysiological monitoring in patients with delayed cerebral ischemia after clipping of ruptured arterial aneurysms. Neyrokhirurgiya = Russian Journal of Neurosurgery. 2024;26(2):26-36. (In Russ.) https://doi.org/10.17650/1683-3295-2024-26-2-26-36
- Rosenthal E.S. The utility of EEG, SSEP, and other neurophysiologic tools to guide neurocritical care. Neurotherapeutics. 2012;9(1):24-36. PMID: 22234455; PMCID: PMC3271154. https://doi.org/10.1007/s13311-011-0101-x
- Fridman J., John E.R., Bergelson M., Kaiser J.B., Baird H.W. Application of digital filtering and automatic peak detection to brain stem auditory evoked potential. Electroencephalogr Clin Neurophysiol. 1982;53(4):405-416. PMID: 6175502. https://doi.org/10.1016/0013-4694(82)90005-0
- Hammerschlag P.E., Berg H.M., Prichep L.S., John E.R., Cohen N.L., Ransohoff J. Real‐time monitoring of brainstem auditory evoked response (BAER) during cerebellopontine angle (CPA) surgery. Otolaryngol Head Neck Surg. 1986;95(5):538-542. PMID: 3108791. https://doi.org/10.1177/019459988609500503
- Toleikis J.R., Pace C., Jahangiri F.R., Hemmer L.B., Toleikis S.C. Intraoperative somatosensory evoked potential (SEP) monitoring: an updated position statement by the American Society of Neurophysiological Monitoring. J Clin Monit Comput. 2024;38(5):1003-1042. PMID: 39068294; PMCID: PMC11427520. https://doi.org/10.1007/s10877-024-01201-x
- Левин Е.А., Кильчуков М.Г., Глушаева А.А. Интраоперационный мониторинг зрительных вызванных потенциалов: опыт 240 операций. Нейрохирургия. 2024;26(3):57-71. https://doi.org/10.17650/1683-3295-2024-26-3-57-71 Levin E.A., Kilchukov M.G., Glushaeva A.A. Intraoperative monitoring of visual evoked potentials: experience of 240 operations. Neyrokhirurgiya = Russian Journal of Neurosurgery. 2024;26(3):57-71. (In Russ.) https://doi.org/10.17650/1683-3295-2024-26-3-57-71
- Kodama K., Goto T., Sato A., Sakai K., Tanaka Y., Hongo K. Standard and limitation of intraoperative monitoring of the visual evoked potential. Acta Neurochir (Wien). 2010;152(4):643-648. PMID: 20127123. https://doi.org/10.1007/s00701-010-0600-2
- Legatt A.D. Chapter 23 — Brainstem auditory evoked potentials (BAEPs) and intraoperative BAEP monitoring. In: Handbook of Clinical Neurophysiology. 2010;9:282-302. https://doi.org/10.1016/S1567-4231(10)09023-4
- MacDonald D.B., Dong C., Quatrale R., Sala F., Skinner S., Soto F., Szelenyi A. Recommendations of the International Society of Intraoperative Neurophysiology for intraoperative somatosensory evoked potentials. Clin Neurophysiol. 2019;130(1):161-179. PMID: 30470625. https://doi.org/10.1016/j.clinph.2018.10.008
- Васильев К.К. Дискретный фильтр Винера. Вестник Ульяновского государственного технического университета. 2016;1(73):47-53. Vasilyev K.K. Discrete filter of Wiener. Vestnik Ulyanovskogo gosudarstvennogo tekhnicheskogo universiteta = Bulletin of Ulyanovsk State Technical University. 2016;1(73):47-53. (In Russ.)

