Опубликован 27.12.2024
Ключевые слова
- натрийуретический пептид,
- полиморфные варианты гена,
- приобретенный порок сердца,
- ревматическая болезнь сердца,
- фибрилляция предсердий
Как цитировать
Copyright (c) 2024 Синицкая А.В., Хуторная М.В., Хрячкова О.Н., Поддубняк А.О., Асанов М.А., Синицкий М.Ю.

Это произведение доступно по лицензии Creative Commons «Attribution» («Атрибуция») 4.0 Всемирная.
Аннотация
Актуальность. Одна из самых распространенных аритмий у пациентов с ревматической болезнью сердца — фибрилляция предсердий. Патофизиологические механизмы ее развития на фоне ревматической болезни сердца не до конца изучены. Как и большинство патологий сердечно-сосудистого континуума, фибрилляция предсердий имеет мультифакториальную природу со значительным вкладом генетической составляющей.
Цель. Поиск ассоциаций полиморфных вариантов генов натрийуретических пептидов (NPPA, NPPB, NPPC) с риском фибрилляции предсердий у пациентов с ревматической болезнью сердца.
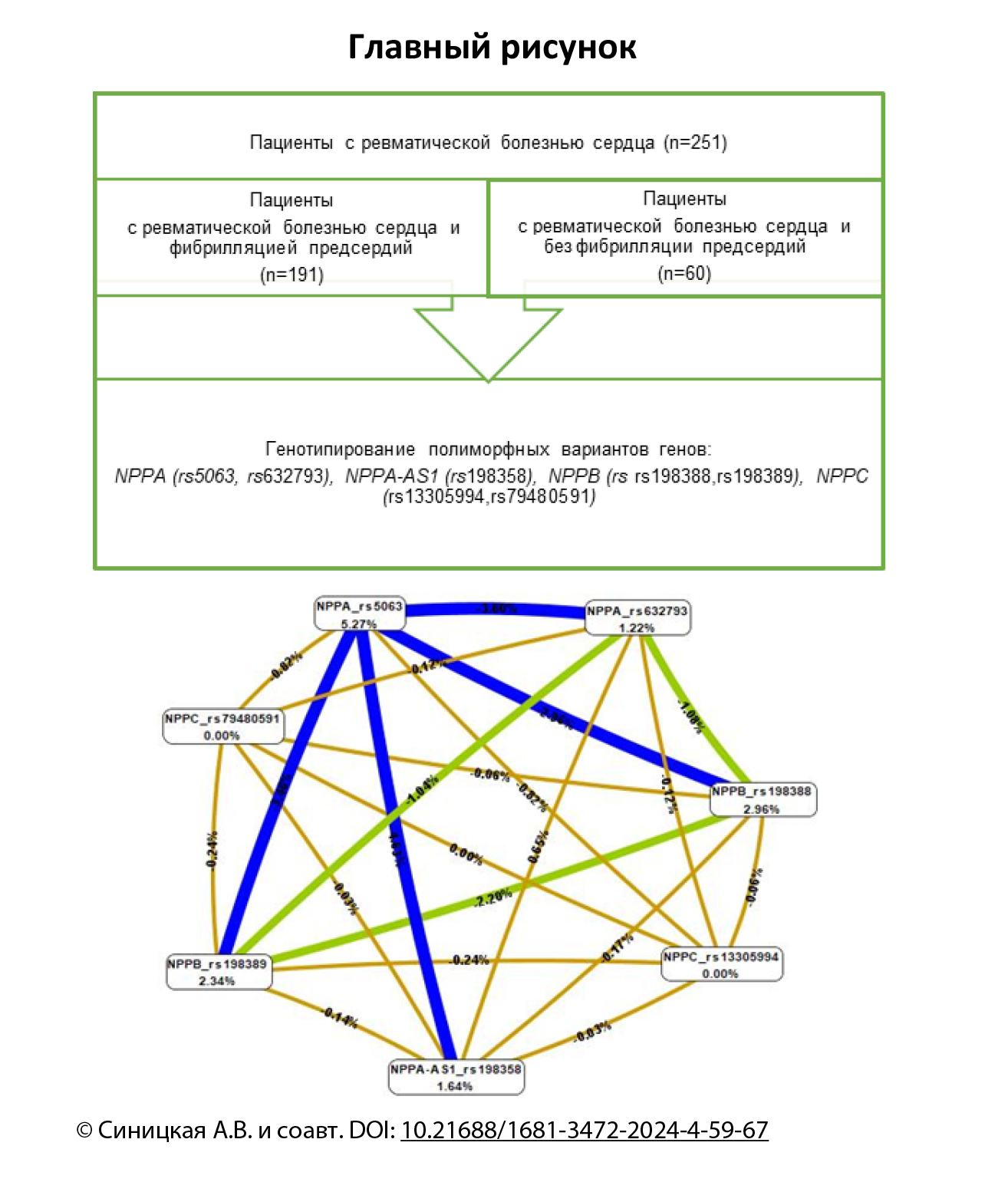
Методы. В исследование включили 251 пациента с диагнозом «ревматическая болезнь сердца». Участников разделили на 2 группы: пациенты с ревматической болезнью сердца и фибрилляцией предсердий (n = 191) и лица с ревматической болезнью сердца без фибрилляции предсердий (n = 60). Генотипирование 7 аллельных вариантов генов, кодирующих натрийуретические пептиды, проводили методом полимеразной цепной реакции в режиме реального времени.
Результаты. Для полиморфных вариантов rs198388 и rs198389 гена NPPB установили протективный эффект в отношении фибрилляции предсердий у пациентов с ревматической болезнью сердца по кодоминантной модели наследования. Аллельный вариант rs198358 гена NPPA-AS1 увеличивает риск фибрилляции предсердий у больных ревматической болезнью сердца в 2 раза (отношение шансов 1,96, 95% доверительный интервал 1,02–3,75, p = 0,037). Выявили 2 модели межгенных взаимодействий, обладающие наибольшими эффективностью и чувствительностью. Наиболее значимыми комбинациями генотипов, ассоциированных с повышенным риском фибрилляции предсердий у пациентов с ревматической болезнью сердца, были rs198388 C/T × rs198389 A/G × rs198358 T/C × rs5063 C/C × rs632793 A/G и rs198388 C/T × rs198358 T/C × rs5063 C/C.
Заключение. Выявили ассоциации полиморфных вариантов генов натрийуретических пептидов (NPPB rs198388, rs198389 и NPPA-AS1 rs198358) с фибрилляцией предсердий у пациентов с ревматической болезнью сердца. Определили трехлокусную (NPPB rs198388, NPPA-AS1 rs198358, NPPA rs5063) и пятилокусную (NPPB rs198388, NPPB rs198389, NPPA-AS1 rs198358, NPPA rs5063, NPPA rs632793) модели взаимодействий генов, ассоциированные с изучаемым фенотипом.
Поступила в редакцию 16 апреля 2024 г. Исправлена 10 октября 2024 г. Принята к печати 2 ноября 2024 г.
Финансирование
Исследование выполнено при поддержке комплексной программы фундаментальных научных исследований СО РАН в рамках фундаментальной темы НИИ КПССЗ № 0419-2022-0001.
Конфликт интересов
Авторы заявляют об отсутствии конфликта интересов.
Вклад авторов
Концепция и дизайн работы: А.В. Синицкая, М.В. Хуторная
Сбор и анализ данных, проведение генотипирования: О.Н. Хрячкова, А.О. Поддубняк, М.В. Хуторная, М.А. Асанов
Статистическая обработка данных: А.В. Синицкая
Написание статьи: А.В. Синицкая
Исправление статьи: М.Ю. Синицкий
Утверждение окончательного варианта статьи: все авторы
Библиографические ссылки
- Дрень Е.В., Ляпина И.Н., Печерина Т.Б., Барбараш О.Л. Фенотип современного пациента с приобретенными пороками клапанов сердца: обзор литературы. CardioСоматика. 2023;14(4):269-282. https://doi.org/10.17816/CS601825 Dren E.V., Lyapina I.N., Pecherina T.B., Barbarash O.L. Phenotype of a modern patient with valvular heart diseases: literature review. CardioSomatics. 2023;14(4):269-282. (In Russ.) https://doi.org/10.17816/CS601825
- Passos L.S.A., Nunes M.C.P., Aikawa E. Rheumatic heart valve disease pathophysiology and underlying mechanisms. Front Cardiovasc Med. 2021;7:612716. PMID: 33537348; PMCID: PMC7848031. https://doi.org/10.3389/fcvm.2020.612716
- Noubiap J.J., Nyaga U.F., Ndoadoumgue A.L., Nkeck J.R., Ngouo A., Bigna J.J. Meta-analysis of the incidence, prevalence, and correlates of atrial fibrillation in rheumatic heart disease. Glob Heart. 2020;15(1):38. PMID: 32923332; PMCID: PMC7427678. https://doi.org/10.5334/gh.807
- Khurshid S., Healey J.S., McIntyre W.F., Lubitz S.A. Population-based screening for atrial fibrillation. Circ Res. 2020;127(1):143-154. PMID: 32716713; PMCID: PMC7388078. https://doi.org/10.1161/circresaha.120.316341
- Jonmundsson T., Steindorsdottir A.E., Austin T.R., Frick E.A., Axelsson G.T., Launer L., Psaty B.M., Loureiro J., Orth A.P., Aspelund T., Emilsson V., Floyd J.S., Jennings L., Gudnason V., Gudmundsdottir V. A proteomic analysis of atrial fibrillation in a prospective longitudinal cohort (AGES-Reykjavik study). Europace. 2023;25(11):euad320. PMID: 37967346; PMCID: PMC10685397. https://doi.org/10.1093/europace/euad320
- Rubattu S., Volpe M. Natriuretic peptides in the cardiovascular system: multifaceted roles in physiology, pathology and therapeutics. Int J Mol Sci. 2019;20(16):3991. PMID: 31426320; PMCID: PMC6721730. https://doi.org/10.3390/ijms20163991
- Guler A., Turkmen I., Atmaca S., Karakurt H., Kahraman S., Aydin S., Sevinc S., Tukenmez Karakurt S., Turkvatan Cansever A., Erturk M., Babur Guler G. Influence of cardiac biomarkers on predicting significant coronary artery disease in hypertrophic cardiomyopathy patients. Heart Vessels. 2023;38(11):1329-1336. PMID: 37414867. https://doi.org/10.1007/s00380-023-02287-0
- Fuery M.A., Leifer E.S., Samsky M.D., Sen S., O'Connor C.M., Fiuzat M., Ezekowitz J., Piña I., Whellan D., Mark D., Felker G.M., Desai N.R., Januzzi J.L., Ahmad T. Prognostic impact of repeated NT-proBNP measurements in patients with heart failure with reduced ejection fraction. JACC Heart Fail. 2024;12(3):479-487. PMID: 38127049. https://doi.org/10.1016/j.jchf.2023.11.007
- Hahn M., Stamer U.M., Luedi M.M., Book M., Rieder H.U., Stüber F. ASA Status, NPPA/NPPB haplotype and coronary artery disease have an impact on BNP/NT-proBNP plasma levels. Cells. 2022;11(5):766. PMID: 35269388; PMCID: PMC8909668. https://doi.org/10.3390/cells11050766
- Rubattu S., Stanzione R., Cotugno M., Bianchi F., Marchitti S., Forte M. Epigenetic control of natriuretic peptides: implications for health and disease. Cell Mol Life Sci. 2020;77(24):5121-5130. PMID: 32556416; PMCID: PMC11105024. https://doi.org/10.1007/s00018-020-03573-0
- Ellis K.L., Newton-Cheh C., Wang T.J., Frampton C.M., Doughty R.N., Whalley G.A., Ellis C.J., Skelton L., Davis N., Yandle T.G., Troughton R.W., Richards A.M., Cameron V.A. Association of genetic variation in the natriuretic peptide system with cardiovascular outcomes. J Mol Cell Cardiol. 2011;50(4):695-701. PMID: 21276798. https://doi.org/10.1016/j.yjmcc.2011.01.010
- Seidelmann S.B., Vardeny O., Claggett B., Yu B., Shah A.M., Ballantyne C.M., Selvin E., MacRae C.A., Boerwinkle E., Solomon S.D. An NPPB promoter polymorphism associated with elevated N-terminal pro-B-type natriuretic peptide and lower blood pressure, hypertension, and mortality. J Am Heart Assoc. 2017;6(4):e005257. PMID: 28341776; PMCID: PMC5533018. https://doi.org/10.1161/JAHA.116.005257
- Кутихин А.Г., Южалин А.Е., Понасенко А.В. Современные тенденции статистической обработки данных и представления результатов в кандидатных генетико-эпидемиологических исследованиях. Фундаментальная и клиническая медицина. 2017;2(2):77-82. https://doi.org/10.23946/2500-0764-2017-2-2-77-82 Kutikhin A.G., Yuzhalin A.E., Ponasenko A.V. How to analyze and present genetic epidemiology data in candidate studies. Fundamental and Clinical Medicine. 2017;2(2):77-82. (In Russ.) https://doi.org/10.23946/2500-0764-2017-2-2-77-82
- Жорина Ю.В., Абрамовских О.С., Игнатова Г.Л. Анализ межгенных взаимодействий полиморфных локусов генов IL4 (C-589T), IL10 (G-1082A; C-592A), IL13 (ARG130GLN) у взрослых больных атопической бронхиальной астмой. Южно-Уральский медицинский журнал. 2020;1:19-31. Zhorina Y.V., Abramovskikh O.S., Ignatova G.L. Analysis of gene-gene interaction between polymorphisms of genes IL4 (C-589T), IL10 (G-1082A; C-592A), IL13 (ARG130GLN) in adult patients with atopic bronchial asthma. South Ural Medical Journal. 2020;1:19-31. (In Russ.)
- Ritchie M.D., Hahn L.W., Roodi N., Bailey L.R., Dupont W.D., Parl F.F., Moore J.H. Multifactor-dimensionality reduction reveals high-order interactions among estrogen-metabolism genes in sporadic breast cancer. Am J Hum Genet. 2001;69(1):138-147. PMID: 11404819; PMCID: PMC1226028. https://doi.org/10.1086/321276
- Пономаренко И.В. Использование метода Multifactor Dimensionality Reduction (MDR) и его модификаций для анализа ген-генных и ген-средовых взаимодействий при генетико-эпидемиологических исследованиях (обзор). Научные результаты биомедицинских исследований. 2019;5(1):4-21. https://doi.org/10.18413/2313-8955-2019-5-1-0-1 Ponomarenko I.V. Using the method of Multifactor Dimensionality Reduction (MDR) and its modifications for analysis of gene-gene and gene-environment interactions in genetic-epidemiological studies (review). Research Results in Biomedicine. 2019;5(1):4-21. (In Russ.) https://doi.org/10.18413/2313-8955-2019-5-1-0-1
- Butt H.I., Shahbaz A., Nawaz H., Butt K. Comparative clinical characteristics of rheumatic heart disease patients undergoing surgical valve replacement. Cureus. 2019;11(6):e4889. PMID: 31423369; PMCID: PMC6689495. https://doi.org/10.7759/cureus.4889
- Bansal A., Sarkar P.G., Chaturvedi V. Atrial fibrillation in rheumatic heart disease. Current Treatment Options in Cardiovascular Medicine. 2020;22(11):42. https://doi.org/10.1007/s11936-020-00845-7
- Pourafkari L., Ghaffari S., Bancroft G.R., Tajlil A., Nader N.D. Factors associated with atrial fibrillation in rheumatic mitral stenosis. Asian Cardiovasc Thorac Ann. 2015;23(1):17-23. PMID: 24696100. https://doi.org/10.1177/0218492314530134
- Huang Y., Wang L.-L., Liu Z.-B., Chen C., Ren X., Luo A.-T., Ma J.-H., Antzelevitch C., Barajas-Martínez H., Hu D. Underlying mechanism of atrial fibrillation-associated Nppa-I137T mutation and cardiac effect of potential drug therapy. Heart Rhythm. 2024;21(2):184-196. PMID: 37924963. https://doi.org/10.1016/j.hrthm.2023.10.025
- Kany S., Reissmann B., Metzner A., Kirchhof P., Darbar D., Schnabel R.B. Genetics of atrial fibrillation — practical applications for clinical management: if not now, when and how? Cardiovasc Res. 2021;117(7):1718-1731. PMID: 33982075; PMCID: PMC8208749. https://doi.org/10.1093/cvr/cvab153
- Goetze J.P., Bruneau B.G., Ramos H.R., Ogawa T., de Bold M.K., de Bold A.J. Cardiac natriuretic peptides. Nat Rev Cardiol. 2020;17(11):698-717. PMID: 32444692. https://doi.org/10.1038/s41569-020-0381-0
- Kerkelä R., Ulvila J., Magga J. Natriuretic peptides in the regulation of cardiovascular physiology and metabolic events. J Am Heart Assoc. 2015;4(10):e002423. PMID: 26508744; PMCID: PMC4845118. https://doi.org/10.1161/JAHA.115.002423
- Ren X., Xu C., Zhan C., Yang Y., Shi L., Wang F., Wang C., Xia Y., Yang B., Wu G., Wang P., Li X., Wang D., Xiong X., Liu J., Liu Y., Liu M., Liu J., Tu X., Wang Q.K. Identification of NPPA variants associated with atrial fibrillation in a Chinese GeneID population. Clin Chim Acta. 2010;411(7-8):481-485. PMID: 20064500. https://doi.org/10.1016/j.cca.2009.12.019
- Olesen M.S., Andreasen L., Jabbari J., Refsgaard L., Haunsø S., Olesen S.P., Nielsen J.B., Schmitt N., Svendsen J.H. Very early-onset lone atrial fibrillation patients have a high prevalence of rare variants in genes previously associated with atrial fibrillation. Heart Rhythm. 2014;11(2):246-251. PMID: 24144883. https://doi.org/10.1016/j.hrthm.2013.10.034
- Silva Cunha P., Antunes D.O., Laranjo S., Coutinho A., Abecasis J., Oliveira M.M. Case report: Mutation in NPPA gene as a cause of fibrotic atrial myopathy. Front Cardiovasc Med. 2023;10:1149717. PMID: 37363091; PMCID: PMC10285104. https://doi.org/10.3389/fcvm.2023.1149717
- Чаулин А.М., Дупляков Д.В. Повышение натрийуретических пептидов, не ассоциированное с сердечной недостаточностью. Российский кардиологический журнал. 2020;25(4S):4140. https://doi.org/10.15829/1560-4071-2020-4140 Chaulin A.M., Duplyakov D.V. Increased natriuretic peptides not associated with heart failure. Rossijskij kardiologicheskij zhurnal = Russian Journal of Cardiology. 2020;25(4S):4140. (In Russ.) https://doi.org/10.15829/1560-4071-2020-4140
- Sarzani R., Allevi M., Di Pentima C., Schiavi P., Spannella F., Giulietti F. Role of cardiac natriuretic peptides in heart structure and function. Int J Mol Sci. 2022;23(22):14415. PMID: 36430893; PMCID: PMC9697447. https://doi.org/10.3390/ijms232214415
- Yanagisawa S., Inden Y., Kato H., Fujii A., Mizutani Y., Ito T., Kamikubo Y., Kanzaki Y., Hirai M., Murohara T. Decrease in B-type natriuretic peptide levels and successful catheter ablation for atrial fibrillation in patients with heart failure. Pacing Clin Electrophysiol. 2016;39(3):225-234. PMID: 26596862. https://doi.org/10.1111/pace.12788
- Çubukçuoğlu Deniz G., Durdu S., Doğan Y., Erdemli E., Özdağ H., Akar A.R. Molecular signatures of human chronic atrial fibrillation in primary mitral regurgitation. Cardiovasc Ther. 2021;2021:5516185. PMID: 34737791; PMCID: PMC8538404. https://doi.org/10.1155/2021/5516185
- Fu S., Ping P., Wang F., Luo L. Synthesis, secretion, function, metabolism and application of natriuretic peptides in heart failure. J Biol Eng. 2018;12:1-21. PMID: 29344085; PMCID: PMC5766980. https://doi.org/10.1186/s13036-017-0093-0
- Johansson Å., Eriksson N., Lindholm D., Varenhorst C., James S., Syvänen A.-C., Axelsson T., Siegbahn A., Barratt B.J., Becker R.C., Himmelmann A., Katus H.A., Steg P.G., Storey R.F., Wallentin L., PLATO Investigators. Genome-wide association and Mendelian randomization study of NT-proBNP in patients with acute coronary syndrome. Hum Mol Genet. 2016;25(7):1447-1456. PMID: 26908625. https://doi.org/10.1093/hmg/ddw012

