Оценка оригинального герметизирующего покрытия с антибактериальным эффектом для синтетических сосудистых протезов
Опубликован 30.03.2023
Ключевые слова
- водопроницаемость,
- желатин,
- сосудистый протез
Как цитировать
Copyright (c) 2023 Шаданов А.А., Журавлева И.Ю., Самойлова Л.М., Тимченко Т.П., Владимиров С.В., Карпова Е.В., Лучников Н.Е., Сирота Д.А., Едемский А.Г., Богачев-Прокофьев А.В., Чернявский А.М.

Это произведение доступно по лицензии Creative Commons «Attribution» («Атрибуция») 4.0 Всемирная.
Аннотация
Актуальность. Инфицирование сосудистого протеза является серьезным осложнением в сердечно-сосудистой хирургии, характеризуется тяжелым течением и высокой смертностью. Несмотря на то что антибактериальные покрытия для сосудистых протезов широко распространены, в мире продолжаются работы по их улучшению.
Цель. Разработка и экспериментальное изучение свойств герметизирующего покрытия с антимикробным эффектом для синтетических сосудистых протезов из дакрона.
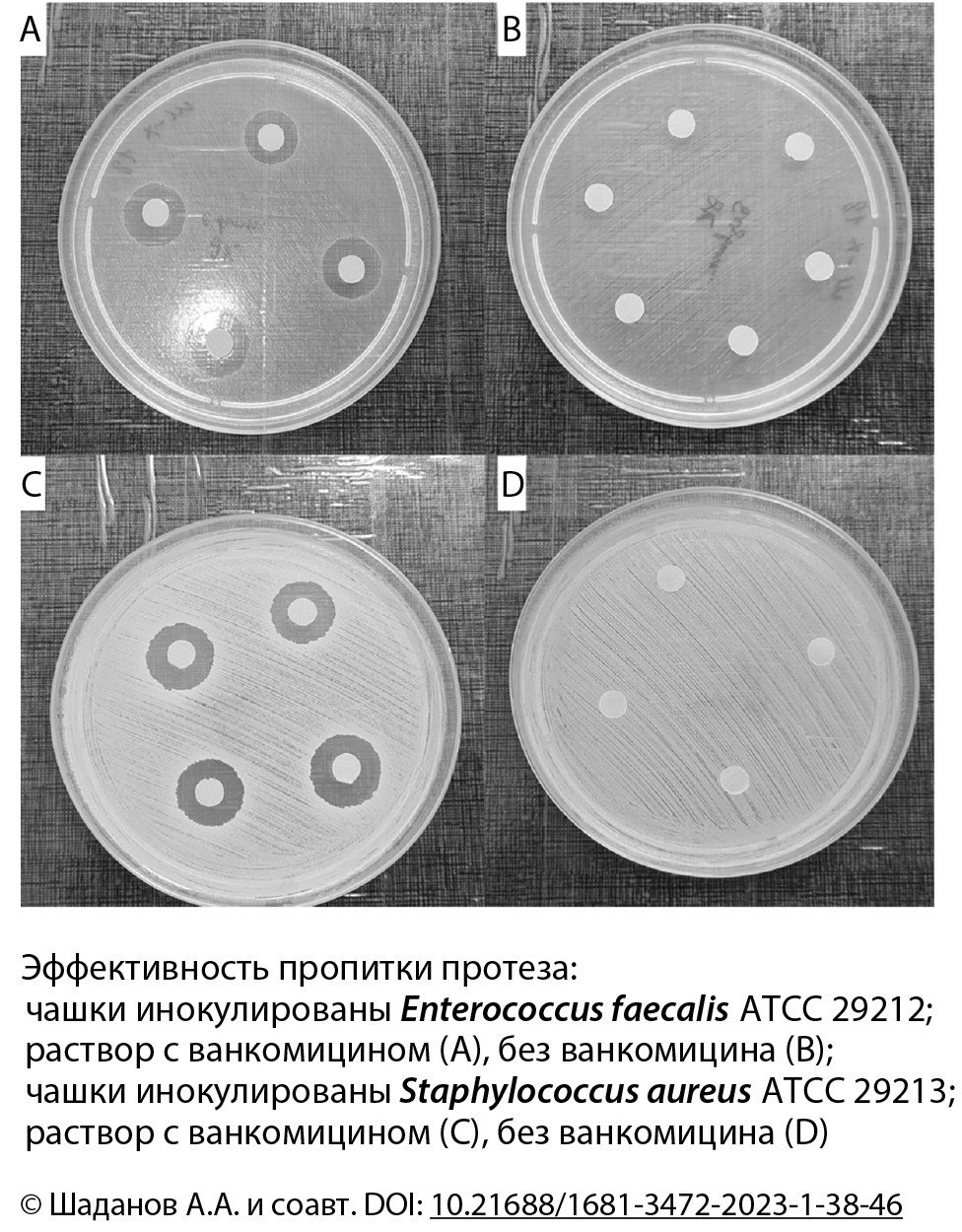
Методы. Использовали 2 типа сосудистых протезов: протез № 1 (полотняный ткацкий раппорт) с исходным уровнем водопроницаемости 78,8 ± 2,7 мл/см2/мин, протез № 2 (саржа 2/1 + атлас 6/4) — уровень водопроницаемости 549,8 ± 20,7 мл/см2/мин. Разработали оригинальную методику желатиновой пропитки, выполнили сканирующую электронную микроскопию, тесты на водопроницаемость и устойчивость к радиальному перегибу. Антибактериальную активность желатиновой пропитки с ванкомицином оценили диско-диффузионным методом на средах с Staphylococcus aureus ATCC 29213 и Enterococcus faecalis ATCC 29212 путем измерения зон задержки роста микроорганизмов после 24-часовой инкубации. Для этого использовали образцы, выкроенные из протезов с пропиткой, содержащей ванкомицин (опытная группа) и не содержащей ванкомицин (контрольная группа).
Результаты. Желатиновое покрытие на образцах визуализируется в виде тонкой ровной пленки и обеспечивает нулевую водопроницаемость. Негативным эффектом является увеличение жесткости и, соответственно, радиуса перегиба. Введение ванкомицина в покрытие привело к значительной задержке роста в культурах микроорганизмов: средний диаметр зоны задержки роста в культуре Staphylococcus aureus составил 15,60 ± 0,65 мм, в культуре Enterococcus faecalis — 14,40 ± 0,66 мм. В протезах, не обработанных ванкомицином, не обнаружили зон задержки роста.
Заключение. Обработка желатиновой пропиткой с раствором ванкомицина — простая профилактическая мера для предотвращения роста грамположительных бактерий на материалах сосудистых протезов.
Поступила в редакцию 14 ноября 2022 г. Исправлена 15 декабря 2022 г. Принята к печати 19 декабря 2022 г.
Финансирование
Работа выполнена при финансовой поддержке Российского научного фонда и правительства Новосибирской области (проект № 22-15-20005).
Конфликт интересов
Авторы заявляют об отсутствии конфликта интересов.
Вклад авторов
Концепция и дизайн работы: И.Ю. Журавлева, А.А. Шаданов, Д.А. Сирота
Сбор и анализ данных: А.А. Шаданов, Л.М. Самойлова, С.В. Владимиров, Т.П. Тимченко, Н.Е. Лучников, Е.В. Карпова
Статистическая обработка данных: А.А. Шаданов
Написание статьи: А.А. Шаданов, Л.М. Самойлова, Т.П. Тимченко, А.Г. Едемский
Исправление статьи: Д.А. Сирота, И.Ю. Журавлева, А.В. Богачев-Прокофьев, А.М. Чернявский
Утверждение окончательного варианта статьи: все авторы
Библиографические ссылки
- Chakfé N., Bizonne S.C., Beaufigeau M., Urban E., Cardon A., Doillon C., Le Magnen J.F., Durand B., Kretz J.G. Impregnated polyester arterial prostheses: performance and prospects. Ann Vasc Surg. 1999;13(5):509-523. PMID: 10466995. https://doi.org/10.1007/s100169900291
- Halpert B., De Bakey M.E., Jordan Jr G.L., Henly W.S. The fate of homografts and prostheses of the human aorta. Surg Gynecol Obstet. 1960;111:659-674. PMID: 13710927.
- Humphries A.W., Hawk W.A., Cuthbertson A.M. Arterial prosthesis of collagen-impregnated Dacron tulle. Surgery. 1961;50:947-954. PMID: 14449985.
- Bascom J.U. Gelatin sealing to prevent blood loss from knitted arterial grafts. Surgery. 1961;50:504-512. PMID: 13687562.
- Prager M., Polterauer P., Böhmig H.J., Wagner O., Fügl A., Kretschmer G., Plohner M., Nanobashvili J., Huk I. Collagen versus gelatin-coated Dacron versus stretch polytetrafluoroethylene in abdominal aortic bifurcation graft surgery: Results of a seven-year prospective, randomized multicenter trial. Surgery. 2001;130(3):408-414. PMID: 11562661. https://doi.org/10.1067/msy.2001.115904
- Lacroix H., Boel K., Nevelsteen A., Suy R. Early inflammatory response to gelatin- and collagen-sealed Dacron prostheses. Ann Vasc Surg. 1995;9(2):152-154. PMID: 7786700. https://doi.org/10.1007/bf02139657
- Domurado D., Thomas D., Brown G. A new method for producing proteic coatings. J Biomed Mater Res. 1975;9(1):109-110. PMID: 809444. https://doi.org/10.1002/jbm.820090110
- Wilson W.R., Bower T.C., Creager M.A., Amin-Hanjani S., O’Gara P.T., Lockhart P.B., Darouiche R.O., Ramlawi B., Derdeyn C.P., Bolger A.F., Levison M.E., Taubert K.A., Baltimore R.S., Baddour L.M.; American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Surgery and Anesthesia; Council on Peripheral Vascular Disease; and Stroke Council. Vascular graft infections, mycotic aneurysms, and endovascular infections: a scientific statement from the American Heart Association. Circulation. 2016;134(20):e412-e460. PMID: 27737955. https://doi.org/10.1161/cir.0000000000000457
- Hall-Stoodley L., Costerton J.W., Stoodley P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2004;2(2):95-108. PMID: 15040259. https://doi.org/10.1038/nrmicro821
- Gandelman G., Frishman W.H., Wiese C., Green-Gastwirth V., Hong S., Aronow W.S., Horowitz H.W. Intravascular device infections: epidemiology, diagnosis, and management. Cardiol Rev. 2007;15(1):13-23. PMID: 17172879. https://doi.org/10.1097/01.crd.0000197966.53529.67
- Herten M., Idelevich E.A., Sielker S., Becker K., Scherzinger A.S., Osada N., Torsello G.B., Bisdas T. Vascular graft impregnation with antibiotics: the influence of high concentrations of rifampin, vancomycin, daptomycin, and bacteriophage endolysin hy-133 on viability of vascular cells. Med Sci Monit Basic Res. 2017;23:250-257. PMID: 28652563; PMCID: PMC5498120. https://doi.org/10.12659/msmbr.902879
- Berard X., Puges M., Pinaquy J.-B., Cazanave C., Stecken L., Bordenave L., Pereyre S., M'Zali F. In vitro evidence of improved antimicrobial efficacy of silver and triclosan containing vascular grafts compared with rifampicin soaked grafts. Eur J Vasc Endovasc Surg. 2019;57(3):424-432. PMID: 30301647. https://doi.org/10.1016/j.ejvs.2018.08.053
- Szałapata K., Osińska-Jaroszuk M., Kapral-Piotrowska J., Pawlikowska-Pawlęga B., Łopucki R., Mroczka R., Jarosz-Wilkołazka A. Serine protease inhibitors — new molecules for modification of polymeric biomaterials. Biomolecules. 2020;10(1):82. PMID: 31947983; PMCID: PMC7023003. https://doi.org/10.3390/biom10010082
- Lew W., Moore W. Antibiotic-impregnated grafts for aortic reconstruction. Semin Vasc Surg. 2011;24(4):211-219. PMID: 22230676. https://doi.org/10.1053/j.semvascsurg.2011.10.015
- Saw M.M., Chandler B., Ho K.M. Benefits and risks of using gelatin solution as a plasma expander for perioperative and critically ill patients: A meta-analysis. Anaesth Intensive Care. 2012;40(1):17-32. PMID: 22313061. https://doi.org/10.1177/0310057x1204000104
- Журавлева И.Ю., Ляшенко М.М., Шаданов А.А., Сирота Д.А., Чернявский А.М. Quo vadimus? Фундаментальные проблемы разработки гибридных протезов грудной аорты. Ангиология и сосудистая хирургия. 2021;27(4):103-112. https://doi.org/10.33529/ANGIO2021412 Zhuravleva I.Yu, Lyashenko M.M., Shadanov A.A., Sirota D.A., Chernyavskiy A.M. Quo vadimus? Fundamental problems of developing hybrid prostheses of thoracic aorta. Angiologiia i Sosudistaia Khirurgiia = Angiology and Vascular Surgery. 2021;27(4):103-112. (In Russ.) https://doi.org/10.33529/ANGIO2021412
- Gristina A.G. Biomaterial-centered infection: microbial adhesion versus tissue integration. Science. 1987;237(4822):1588-1595. PMID: 3629258. https://doi.org/10.1126/science.3629258
- Schmitt D.D., Bandyk D.F., Pequet A.J., Towne J.B. Bacterial adherence to vascular prostheses. A determinant of graft infectivity. J Vasc Surg. 1986;3(5):732-740. PMID: 2939263.
- Moore W.S., Chvapil M., Seiffert G., Keown K. Development of an infection-resistant vascular prosthesis. Arch Surg. 1981;116(11):1403-1407. PMID: 6458258. https://doi.org/10.1001/archsurg.1981.01380230027004
- Zilberman M., Elsner J.J. Antibiotic-eluting medical devices for various applications. J Control Release. 2008;130(3):202-215. PMID: 18687500. https://doi.org/10.1016/j.jconrel.2008.05.020
- Chervu A., Moore W.S., Gelabert H.A., Colburn M.D., Chvapil M. Prevention of graft infection by use of prostheses bonded with a rifampin/collagen release system. J Vasc Surg. 1991;14(4):521-525. PMID: 1833564. https://doi.org/10.1016/0741-5214(91)90246-Q
- Galdbart J.O., Branger C., Andreassian B., Lambert-Zechovsky N., Kitzis M. Elution of six antibiotics bonded to polyethylene vascular grafts sealed with three proteins. J Surg Res. 1996;66(2):174-178. PMID: 9024831. https://doi.org/10.1006/jsre.1996.0391
- Goëau-Brissonnière O., Leport C., Bacourt F., Lebrault C., Comte R., Pechère J.C. Prevention of vascular graft infection by rifampin bonding to a gelatin-sealed Dacron graft. Ann Vasc Surg. 1991;5(5):408-412. PMID: 1835641. https://doi.org/10.1007/bf02133043
- Morishima M., Marui A., Yanagi S., Nomura T., Nakajima N., Hyon S.-H., Ikeda T., Sakata R. Sustained release of vancomycin from a new biodegradable glue to prevent methicillin-resistant Staphylococcus aureus graft infection. Interact Cardiovasc Thorac Surg. 2010;11(1):52-55. PMID: 20360210. https://doi.org/10.1510/icvts.2010.232447
- Blanchemain N., Laurent T., Chai F., Neut C., Haulon S., Krump-konvalinkova V., Morcellet M., Martel B., Kirkpatrick C.J., Hildebrand H.F. Polyester vascular prostheses coated with a cyclodextrin polymer and activated with antibiotics: cytotoxicity and microbiological evaluation. Acta Biomater. 2008;4(6):1725-1733. PMID: 18676187. https://doi.org/10.1016/j.actbio.2008.07.001

