Non-pharmacologic nephroprotective strategies combined with nitric oxide delivery in cardiac surgical patients with chronic kidney disease: a randomized controlled trial
Published 2024-12-27
Keywords
- Acute Kidney Injury,
- Cardiopulmonary Bypass,
- Cardiac Surgical Procedures,
- Control Groups,
- Nitric Oxide
- Perfusion,
- Renal Insufficiency, Chronic ...More
How to Cite
Copyright (c) 2024 Tyo M.A., Podoksenov Yu.K., Kravchenko I.V., Churilina E.A., Svirko Yu.S., Kozlov B.N., Kamenshchikov N.O.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Introduction: One of the promising areas of nephroprotection in cardiac surgery is perioperative nitric oxide donation. The combined using of Goal-Directed Perfusion (GDP), Kidney Disease Improving Global Outcomes (KDIGO) and nitric oxide delivery strategies can reduce the incidence of acute kidney injury by targeting different links in the pathogenesis of kidney injury.
Objective: To assess the nephroprotective properties of nitric oxide delivery in combination with KDIGO and GDP strategies during cardiac surgery with cardiopulmonary bypass in patients with chronic kidney disease.
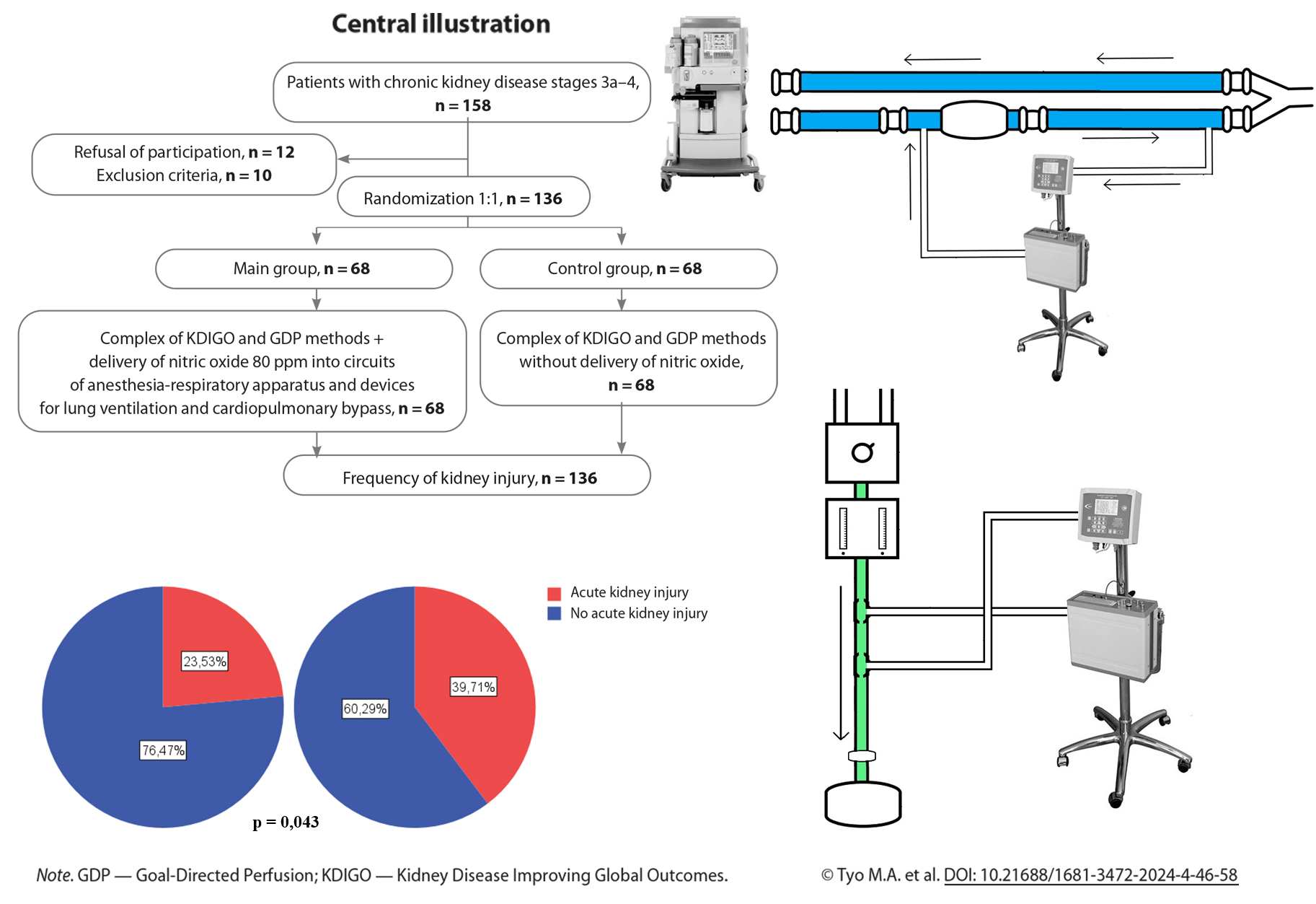
Methods: The study included 136 cardiac surgery patients with chronic kidney disease. The patients were randomized into 2 groups of 68 individuals each. The complex of non-pharmacological nephroprotection methods KDIGO and GDP was used in the control group. In the main group, perioperative delivery of nitric oxide was performed along with a set of KDIGO and GDP measures.
Results: In the main group, the incidence of acute kidney injury was significantly lower compared to the control: 23.5% versus 39.7% (p = 0.043), respectively. Both groups did not differ in the concentration of renal injury biomarkers. In the control group, the concentration of exhaled nitric oxide significantly decreased 2 hours after surgery (p < 0.001), while in the main group no changes were recorded (p = 0.966).
Conclusion: During cardiac surgery in patients with chronic kidney disease, delivery of nitric oxide in combination with the KDIGO and GDP measures reduced the incidence of acute kidney injury compared with the isolated using of non-pharmacological nephroprotective methods via leveling the perioperative deficiency of endogenous nitric oxide but did not affect the expression of kidney injury biomarkers.
ClinicalTrials.gov ID NCT05757557
Received 31 October 2024. Revised 21 November 2024. Accepted 22 November 2024.
Funding
The study was performed within the framework of the state assignment (topic No. 122123000017-3).
Conflict of interest
The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: M.A. Tyo, N.O. Kamenshchikov, Yu.K. Podoksenov, B.N. Kozlov
Data collection and analysis: M.A. Tyo, I.V. Kravchenko, E.A. Churilina, Yu.S. Svirko
Statistical analysis: M.A. Tyo, I.V. Kravchenko
Drafting the article: M.A. Tyo
Critical revision of the article: N.O. Kamenshchikov, Yu.K. Podoksenov, B.N. Kozlov
Final approval of the version to be published: M.A. Tyo, Yu.K. Podoksenov, I.V. Kravchenko, E.A. Churilina, Yu.S. Svirko, B.N. Kozlov, N.O. Kamenshchikov
References
- Kertai M.D., Zhou S., Karhausen J.A., Cooter M., Jooste E., Li Y.-J., White W.D., Aronson S., Podgoreanu M.V., Gaca J., Welsby I.J., Levy J.H., Stafford-Smith M., Mathew J.P., Fontes M.L. Platelet counts, acute kidney injury, and mortality after coronary artery bypass grafting surgery. Anesthesiology. 2016;124(2):339-352. PMID: 26599400; PMCID: PMC5040517. https://doi.org/10.1097/ALN.0000000000000959
- Grams M.E., Sang Y., Coresh J., Ballew S., Matsushita K., Molnar M.Z., Szabo Z., Kalantar-Zadeh K., Kovesdy C.P. Acute kidney injury after major surgery: a retrospective analysis of veterans health administration data. Am J Kidney Dis. 2016;67(6):872-880. PMID: 26337133; PMCID: PMC4775458. https://doi.org/10.1053/j.ajkd.2015.07.022
- Waikar S.S., Curhan G.C., Wald R., McCarthy E.P., Chertow G.M. Declining mortality in patients with acute renal failure, 1988 to 2002. J Am Soc Nephrol. 2006;17(4):1143-1150. PMID: 16495376. https://doi.org/10.1681/ASN.2005091017
- Peng K., McIlroy D.R., Bollen B.A., Billings F.T. 4th, Zarbock A., Popescu W.M., Fox A.A., Shore-Lesserson L., Zhou S., Geube M.A., Ji F., Bhatia M., Schwann N.M., Shaw A.D., Liu H. Society of Cardiovascular Anesthesiologists clinical practice update for management of acute kidney injury associated with cardiac surgery. Anesth Analg. 2022;135(4):744-756. PMID: 35544772. https://doi.org/10.1213/ANE.0000000000006068
- Lei C., Berra L., Rezoagli E., Yu B., Dong H., Yu S., Hou L., Chen M., Chen W., Wang H., Zheng Q., Shen J., Jin Z., Chen T., Zhao R., Christie E., Sabbisetti V.S., Nordio F., Bonventre J.V., Xiong L., Zapol W.M. Nitric oxide decreases acute kidney injury and stage 3 chronic kidney disease after cardiac surgery. Am J Respir Crit Care Med. 2018;198(10):1279-1287. PMID: 29932345; PMCID: PMC6290943. https://doi.org/10.1164/rccm.201710-2150OC
- Kamenshchikov N.O., Anfinogenova Y.J., Kozlov B.N., Svirko Y.S., Pekarskiy S.E., Evtushenko V.V., Lugovsky V.A., Shipulin V.M., Lomivorotov V.V., Podoksenov Y.K. Nitric oxide delivery during cardiopulmonary bypass reduces acute kidney injury: a randomized trial. J Thorac Cardiovasc Surg. 2022;163(4):1393-1403.e9. PMID: 32718702. https://doi.org/10.1016/j.jtcvs.2020.03.182
- Тё М.А., Каменщиков Н.О., Подоксенов Ю.К., Мухомедзянов А.В., Маслов Л.Н., Козлов Б.Н. Влияние доставки оксида азота на энергетическое обеспечение почечной ткани при проведении искусственного кровообращения: экспериментальное исследование. Сибирский журнал клинической и экспериментальной медицины. 2024;39(1):163-170. https://doi.org/10.29001/2073-8552-2022-592 Tyo M.A., Kamenshchikov N.O., Podoksenov Yu.K., Mukhomedzyanov A.V., Maslov L.N., Kozlov B.N. Effect of nitric oxide delivery on energy supply of renal tissue in cardiopulmonary bypass: an experimental study. Siberian Journal of Clinical and Experimental Medicine. 2024;39(1):163-170. (In Russ.) https://doi.org/10.29001/2073-8552-2022-592
- Те М.А., Каменщиков Н.О., Подоксенов Ю.К., Мухомедзянов А.В., Маслов Л.Н., Кравченко И.В., Чурилина Е.А., Козлов Б.Н. Влияние доставки оксида азота на процессы апоптоза, некроптоза и пироптоза в почечной паренхиме при моделировании искусственного кровообращения: экспериментальное исследование. Вестник анестезиологии и реаниматологии. 2024;21(3):26-33. https://doi.org/10.24884/2078-5658-2024-21-3-26-33 Tyo M.A., Kamenshchikov N.O., Podoksenov Yu.K., Mukhomedzyanov A.V., Maslov L.N., Kravchenko I.V., Churilina E.A., Kozlov B.N. The influence of nitric oxide delivery on the processes of apoptosis, necroptosis and pyroptosis in the renal parenchyma after simulating cardiopulmonary bypass: an experimental study. Messenger of Anesthesiology and Resuscitation. 2024;21(3):26-33. (In Russ.) https://doi.org/10.24884/2078-5658-2024-21-3-26-33
- Кравченко И.В., Геренг Е.А., Подоксенов Ю.К., Тё М.А., Серебрякова О.Н., Бянкина М.А., Горохова А.В., Козлов Б.Н., Мильто И.В., Каменщиков Н.О. Влияние доставки оксида азота на морфофункциональное состояние легких при моделировании искусственного кровообращения: экспериментальное исследование. Пульмонология. 2024;34(3):385-394. https://doi.org/10.18093/0869-0189-2024-34-3-385-394 Kravchenko I.V., Gereng E.A., Podoksenov Yu.K., Tyo M.A., Serebryakova O.N., Byankina M.A., Gorokhova A.V., Kozlov B.N., Milto I.V., Kamenshchikov N.O. Effect of nitric oxide supply on the morphofunctional state of the lungs during cardiopulmonary bypass modelling: an experimental study. Pulmonologiya = Pulmonology. 2024;34(3):385-394. (In Russ.) https://doi.org/10.18093/0869-0189-2024-34-3-385-394
- Kamenshchikov N.O., Podoksenov Y.K., Kozlov B.N., Maslov L.N., Mukhomedzyanov A.V., Tyo M.A., Boiko A.M., Margolis N.Y., Boshchenko A.A., Serebryakova O.N., Dzyuman A.N., Shirshin A.S., Buranov S.N., Selemir V.D. The nephroprotective effect of nitric oxide during extracorporeal circulation: an experimental study. Biomedicines. 2024;12(6):1298. PMID: 38927505; PMCID: PMC11201384. https://doi.org/10.3390/biomedicines12061298
- Blum M., Yachnin T., Wollman Y., Chernihovsky T., Peer G., Grosskopf I., Kaplan E., Silverberg D., Cabili S., Iaina A. Low nitric oxide production in patients with chronic renal failure. Nephron. 1998;79(3):265-268. PMID: 9678424. https://doi.org/10.1159/000045047
- Vermeulen Windsant I.C., de Wit N.C.J., Sertorio J.T.C., van Bijnen A.A., Ganushchak Y.M., Heijmans J.H., Tanus-Santos J.E., Jacobs M.J., Maessen J.G., Buurman W.A. Hemolysis during cardiac surgery is associated with increased intravascular nitric oxide consumption and perioperative kidney and intestinal tissue damage. Front Physiol. 2014;5:340. PMID: 25249983; PMCID: PMC4157603. https://doi.org/10.3389/fphys.2014.00340
- KDIGO Board Members. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2012;3(1):1-163. Available from: https://kdigo.org/wp-content/uploads/2017/02/KDIGO_2012_CKD_GL.pdf
- Khwaja A.; KDIGO Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Nephron Clin Pract. 2012;120(4):c179-c184. PMID: 22890468. https://doi.org/10.1159/000339789
- Meersch M., Schmidt C., Hoffmeier A., Van Aken H., Wempe C., Gerss J., Zarbock A. Prevention of cardiac surgery-associated AKI by implementing the KDIGO guidelines in high risk patients identified by biomarkers: the PrevAKI randomized controlled trial. Intensive Care Med. 2017;43(11):1551-1561. PMID: 28110412; PMCID: PMC5633630. https://doi.org/10.1007/s00134-016-4670-3
- Cho J.S., Shim J.-K., Lee S., Song J.-W., Choi N., Lee S., Kwak Y.-L. Chronic progression of cardiac surgery associated acute kidney injury: Intermediary role of acute kidney disease. J Thorac Cardiovasc Surg. 2021;161(2):681-688.e3. PMID: 31959433. https://doi.org/10.1016/j.jtcvs.2019.10.101
- Hu J., Spina S., Zadek F., Kamenshchikov N.O., Bittner E.A., Pedemonte J., Berra L. Effect of nitric oxide on postoperative acute kidney injury in patients who underwent cardiopulmonary bypass: a systematic review and meta-analysis with trial sequential analysis. Ann Intensive Care. 2019;9(1):129. PMID: 31754841; PMCID: PMC6872705. https://doi.org/10.1186/s13613-019-0605-9
- Ranucci M., Pavesi M., Mazza E., Bertucci C., Frigiola A., Menicanti L., Ditta A., Boncilli A., Conti D. Risk factors for renal dysfunction after coronary surgery: the role of cardiopulmonary bypass technique. Perfusion. 1994;9(5):319-326. PMID: 7833539. https://doi.org/10.1177/026765919400900503
- Ranucci M., Romitti F., Isgrò G., Cotza M., Brozzi S., Boncilli A., Ditta A. Oxygen delivery during cardiopulmonary bypass and acute renal failure after coronary operations. Ann Thorac Surg. 2005;80(6):2213-2220. PMID: 16305874. https://doi.org/10.1016/j.athoracsur.2005.05.069
- de Somer F., Mulholland J.W., Bryan M.R., Aloisio T., Van Nooten G.J., Ranucci M. O2 delivery and CO2 production during cardiopulmonary bypass as determinants of acute kidney injury: time for a goal-directed perfusion management? Crit Care. 2011;15(4):R192. PMID: 21831302; PMCID: PMC3387634. https://doi.org/10.1186/cc10349
- Ranucci M., Johnson I., Willcox T., Baker R.A., Boer C., Baumann A., Justison G.A., de Somer F., Exton P., Agarwal S., Parke R., Newland R.F., Haumann R.G., Buchwald D., Weitzel N., Venkateswaran R., Ambrogi F., Pistuddi V. Goal-directed perfusion to reduce acute kidney injury: a randomized trial. J Thorac Cardiovasc Surg. 2018;156(5):1918-1927.e2. PMID: 29778331. https://doi.org/10.1016/j.jtcvs.2018.04.045
- Mukaida H., Matsushita S., Yamamoto T., Minami Y., Sato G., Asai T., Amano A. Oxygen delivery-guided perfusion for the prevention of acute kidney injury: a randomized controlled trial. J Thorac Cardiovasc Surg. 2023;165(2):750-760.e5. PMID: 33840474. https://doi.org/10.1016/j.jtcvs.2021.03.032
- Joannidis M., Druml W., Forni L.G., Groeneveld A.B.J., Honore P.M., Hoste E., Ostermann M., Oudemans-van Straaten H.M., Schetz M. Prevention of acute kidney injury and protection of renal function in the intensive care unit: update 2017: Expert opinion of the Working Group on Prevention, AKI section, European Society of Intensive Care Medicine. Intensive Care Med. 2017;43(6):730-749. PMID: 28577069; PMCID: PMC5487598. https://doi.org/10.1007/s00134-017-4832-y
- Ostermann M., Cennamo A., Meersch M., Kunst G. A narrative review of the impact of surgery and anaesthesia on acute kidney injury. Anaesthesia. 2020;75 Suppl 1:e121-e133. PMID: 31903567. https://doi.org/10.1111/anae.14932
- Yan Y., Kamenshchikov N., Zheng Z., Lei C. Inhaled nitric oxide and postoperative outcomes in cardiac surgery with cardiopulmonary bypass: a systematic review and meta-analysis. Nitric Oxide. 2024;146:64-74. PMID: 38556145. https://doi.org/10.1016/j.niox.2024.03.004
- Kielstein J.T., Böger R.H., Bode-Böger S.M., Schäffer J., Barbey M., Koch K.M., Frölich J.C. Asymmetric dimethylarginine plasma concentrations differ in patients with end-stage renal disease: relationship to treatment method and atherosclerotic disease. J Am Soc Nephrol. 1999;10(3):594-600. PMID: 10073610. https://doi.org/10.1681/ASN.V103594
- Tizianello A., De Ferrari G., Garibotto G., Gurreri G., Robaudo C. Renal metabolism of amino acids and ammonia in subjects with normal renal function and in patients with chronic renal insufficiency. J Clin Invest. 1980;65(5):1162-1173. PMID: 7364943; PMCID: PMC371450. https://doi.org/10.1172/JCI109771
- Peppa M., Uribarri J., Cai W., Lu M., Vlassara H. Glycoxidation and inflammation in renal failure patients. Am J Kidney Dis. 2004;43(4):690-695. PMID: 15042546. https://doi.org/10.1053/j.ajkd.2003.11.022
- Xiao S., Wagner L., Mahaney J., Baylis C. Uremic levels of urea inhibit L-arginine transport in cultured endothelial cells. Am J Physiol Renal Physiol. 2001;280(6):F989-F995. PMID: 11352838; PMCID: PMC2756804. https://doi.org/10.1152/ajprenal.2001.280.6.F989
- Billings F.T. 4th, Ball S.K., Roberts L.J. 2nd, Pretorius M. Postoperative acute kidney injury is associated with hemoglobinemia and an enhanced oxidative stress response. Free Radic Biol Med. 2011;50(11):1480-1487. PMID: 21334433; PMCID: PMC3090463. https://doi.org/10.1016/j.freeradbiomed.2011.02.011
- Reddy Y.S., Kiranmayi V.S., Bitla A.R., Krishna G.S., Srinivasa Rao P.V.L.N., Sivakumar V. Nitric oxide status in patients with chronic kidney disease. Indian J Nephrol. 2015;25(5):287-291. PMID: 26628794; PMCID: PMC4588324. https://doi.org/10.4103/0971-4065.147376
- Lübbers D.W., Baumgärtl H. Heterogeneities and profiles of oxygen pressure in brain and kidney as examples of the pO2 distribution in the living tissue. Kidney Int. 1997;51(2):372-380. PMID: 9027709. https://doi.org/10.1038/ki.1997.49
- Ishibe Y., Liu R., Hirosawa J., Kawamura K., Yamasaki K., Saito N. Exhaled nitric oxide level decreases after cardiopulmonary bypass in adult patients. Crit Care Med. 2000;28(12):3823-3827. PMID: 11153620. https://doi.org/10.1097/00003246-200012000-00012
- Marczin N., Kövesi T., Royston D. Exhaled nitric oxide as a marker of lung injury in coronary artery bypass surgery. Br J Anaesth. 2003;90(1):101-105. PMID: 12488391.
- Корабельников Д.И., Магомедалиев М.О. Современные биомаркеры острого повреждения почек. Фармакоэкономика. Современная фармакоэкономика и фармакоэпидемиология. 2023;16(1):87-104. https://doi.org/10.17749/2070-4909/farmakoekonomika.2023.171 Korabelnikov D.I., Magomedaliev M.O. Modern biomarkers of acute kidney injury. Farmakoekonomika. Modern Pharmacoeconomics and Pharmacoepidemiology. 2023;16(1):87-104. (In Russ.) https://doi.org/10.17749/2070-4909/farmakoekonomika.2023.171
- Греков И.С. Биомаркеры кардиохирургически-ассоциированного острого повреждения почек. Вестник СурГУ. Медицина. 2021;3(49):61-70. https://doi.org/10.34822/2304-9448-2021-3-61-70 Grekov I.S. Biomarkers of cardiac surgery-associated acute kidney injury. Vestnik SurGU. Meditsina = Bulletin of SurGU. Medicine. 2021;3(49):61-70. (In Russ.) https://doi.org/10.34822/2304-9448-2021-3-61-70
- Molnar A.O., Parikh C.R., Coca S.G., Thiessen-Philbrook H., Koyner J.L., Shlipak M.G., Lee Myers M., Garg A.X., TRIBE-AKI Consortium. Association between preoperative statin use and acute kidney injury biomarkers in cardiac surgical procedures. Ann Thorac Surg. 2014;97(6):2081-2087. PMID: 24725831; PMCID: PMC4068122. https://doi.org/10.1016/j.athoracsur.2014.02.033
- Hsu C.-Y., Xie D., Waikar S.S., Bonventre J.V., Zhang X., Sabbisetti V., Mifflin T.E., Coresh J., Diamantidis C.J., He J., Lora C.M., Miller E.R., Nelson R.G., Ojo A.O., Rahman M., Schelling J.R., Wilson F.P., Kimmel P.L., Feldman H.I., Vasan R.S., Liu K.D.; CRIC Study Investigators; CKD Biomarkers Consortium. Urine biomarkers of tubular injury do not improve on the clinical model predicting chronic kidney disease progression. Kidney Int. 2017;91(1):196-203. PMID: 28029431; PMCID: PMC5362331. https://doi.org/10.1016/j.kint.2016.09.003

