Implantation of a self-expandable prototype transcatheter pulmonary valve in a pig model: an experimental study
Published 2024-11-08
Keywords
- Animals,
- Bioprosthesis,
- Heart Defects, Congenital,
- Pulmonary Artery,
- Pulmonary Valve
- Swine ...More
How to Cite
Copyright (c) 2024 Soynov I.A., Rzaeva K.A., Manukian S.N., Vladimirov S.V., Dokuchaeva A.A., Gorbatykh A.V., Nichai N.R., Amansahatova E.N., Kulyabin Yu.Yu., Magbulova S.A., Zhuravleva I.Yu.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Introduction: Reconstruction of the right ventricular outflow tract is recommended for 20% of patients with congenital heart defects. The risks of complications during repeated open cardiac surgeries stimulated the implantation of the pulmonary artery valve according to the P. Bonhoeffer method developed more than 20 years ago. Modern transcatheter technologies enable implantation of the pulmonary artery valve in patients after transannular plastic surgery in combination with pulmonary regurgitation and not only when using biological valve conduits.
Objective: The aim of this study was to evaluate the short- and mid-term outcomes of implanting a prototype of a self-expanding, transcatheter pulmonary artery valve in an experimental pig model. This study assesses the efficacy and safety of the novel device in comparison to traditional surgical methods.
Methods: A total of 8 experimental animals were included in this study. The object of the study was a transcatheter self-expandable valve-containing prosthetic device designed for implantation in the pulmonary artery, which consisted of an external supporting mesh scaffold made of a titanium nickelide as well as sheathing and a leaflet mechanism.
Results: In the early postoperative period, there was a minor complication in the form of a 1.5 cm small paraprosthetic fistula, which spontaneously closed in the long-term. No mortality or other complications were observed in the mid-term follow-up period. We also noted an insignificant increase in transvalvular gradients, with a gradient of 11 (8-13) mm Hg after surgery, and 16 (12-19) mm Hg in 6 months, p = 0.79. Upon removal animals from the experiment, the supporting scaffolds of the valves were intact without any damage. The valve surface was smooth, without traces of blood clots or cellular infiltration. Microscopically, there was no evidence of structural injuries and no signs of calcification after von Kossa staining.
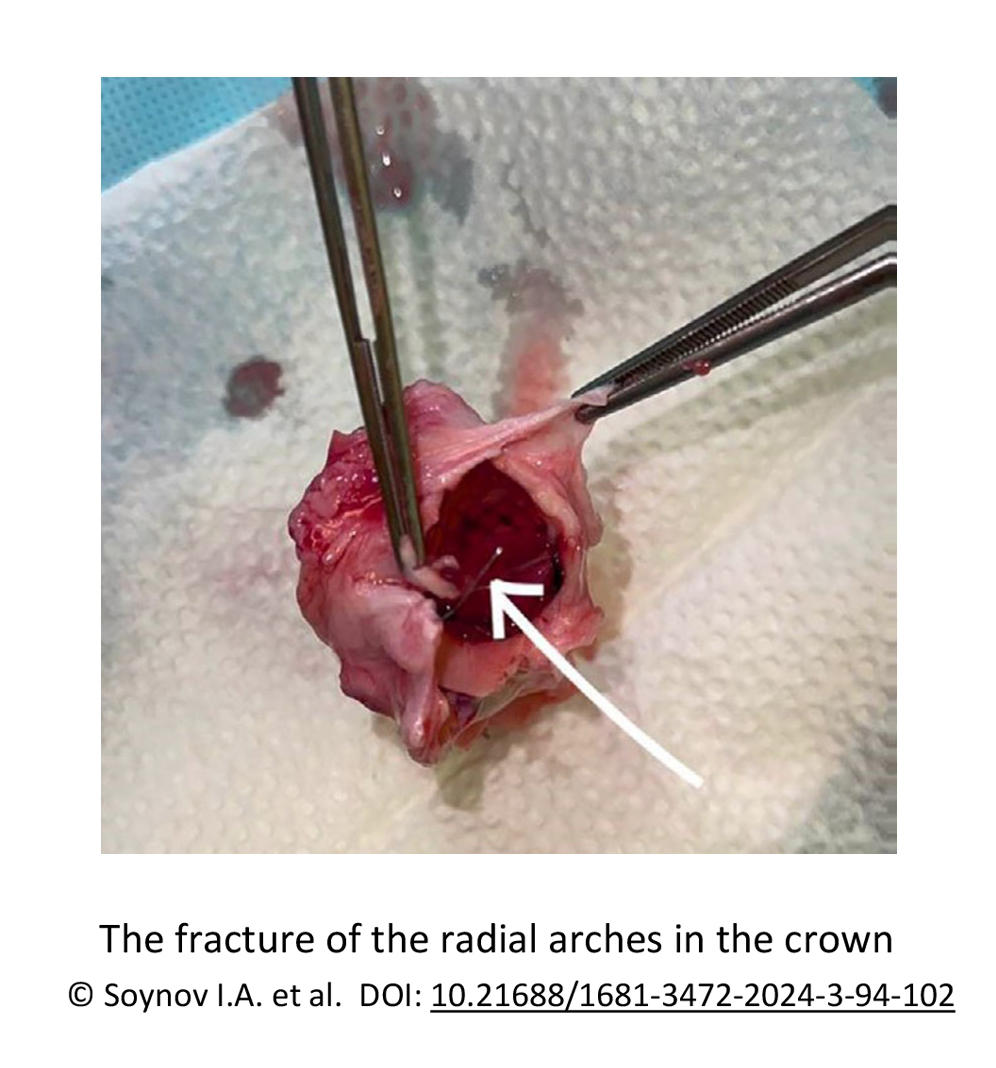
Conclusion: The experimental transcatheter-implanted valve demonstrated good short- and mid-term efficacy without significant pulmonary insufficiency signs and pressure gradient on the bioprosthesis. However, the fracture of the radial arches in the crown of the device, which occurred in 25% of cases, attests the demand on creating a more durable structure through reinforcement of rigidity or increasing the number of radial arches.
Received 27 September 2024. Revised 24 October 2024. Accepted 25 October 2024.
Funding
The research was carried out within the framework of the state assignment of the Ministry of Health of the Russian Federation (No. 124022500251-0).
Conflict of interest
The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: I.A. Soynov, K.A. Rzaeva, S.N. Manukian, S.V. Vladimirov, A.A. Dokuchaeva
Data collection and analysis: A.V. Gorbatykh, N.R. Nichai, E.N. Amansahatova, Yu.Yu. Kulyabin, S.A. Magbulova
Statistical analysis: I.Yu. Zhuravleva, I.A. Soynov, K.A. Rzaeva, S.N. Manukian, S.V. Vladimirov, A.A. Dokuchaeva
Drafting the article: I.A. Soynov, K.A. Rzaeva, S.N. Manukian, S.V. Vladimirov, A.A. Dokuchaeva, A.V. Gorbatykh, N.R. Nichai
Critical revision of the article: I.Yu. Zhuravleva, A.A. Dokuchaeva, A.V. Gorbatykh, N.R. Nichai
Final approval of the version to be published: I.A. Soynov, K.A. Rzaeva, S.N. Manukian, S.V. Vladimirov, A.A. Dokuchaeva, A.V. Gorbatykh, N.R. Nichai, E.N. Amansahatova, Yu.Yu. Kulyabin, S.A. Magbulova, I.Yu. Zhuravleva
References
- Matoq A., Shahanavaz S. Transcatheter pulmonary valve in congenital heart disease. Interv Cardiol Clin. 2024;13(3):369-384. PMID: 38839170. https://doi.org/10.1016/j.iccl.2024.03.001
- Giugno L., Faccini A., Carminati M. Percutaneous pulmonary valve implantation. Korean Circ J. 2020;50(4):302-316. PMID: 32157831; PMCID: PMC7067602. https://doi.org/10.4070/kcj.2019.0291
- Baumgartner H., De Backer J., Babu-Narayan S.V., Budts W., Chessa M., Diller G.-P., Lung B., Kluin J., Lang I.M., Meijboom F., Moons P., Mulder B.J.M., Oechslin E., Roos-Hesselink J.W., Schwerzmann M., Sondergaard L., Zeppenfeld K.; ESC Scientific Document Group. 2020 ESC Guidelines for the management of adult congenital heart disease. Eur Heart J. 2021;42(6):563-645. PMID: 32860028. https://doi.org/10.1093/eurheartj/ehaa554
- Bonhoeffer P., Boudjemline Y., Saliba Z., Hausse A.O., Aggoun Y., Bonnet D., Sidi D., Kachaner J. Transcatheter implantation of a bovine valve in pulmonary position: a lamb study. Circulation. 2000;102(7):813-816. PMID: 10942752. https://doi.org/10.1161/01.cir.102.7.813
- Law M.A., Chatterjee A. Transcatheter pulmonic valve implantation: techniques, current roles, and future implications. World J Cardiol. 2021;13(5):117-129. PMID: 34131475; PMCID: PMC8173335. https://doi.org/10.4330/wjc.v13.i5.117
- Ribeiro J.M., Teixeira R., Lopes J., Costa M., Pires A., Gonçalves L. Transcatheter versus surgical pulmonary valve replacement: a systemic review and meta-analysis. Ann Thorac Surg. 2020;110(5):1751-1761. PMID: 32268142. https://doi.org/10.1016/j.athoracsur.2020.03.007
- Chau A.K.-T. Transcatheter pulmonary valve replacement in congenital heart diseases. Pediatr Investig. 2022;6(4):280-290. PMID: 36582274; PMCID: PMC9789934. https://doi.org/10.1002/ped4.12359
- Patel N.D., Levi D.S., Cheatham J.P., Qureshi S.A., Shahanavaz S., Zahn E.M. Transcatheter pulmonary valve replacement: a review of current valve technologies. J Soc Cardiovasc Angiogr Interv. 2022;1(6):100452. PMID: 39132347; PMCID: PMC11307711. https://doi.org/10.1016/j.jscai.2022.100452
- Тихонов В.Н., Бобович В.Е., Запорожец В.И. Создание нового поколения супермелких лабораторных свиней для работы в области медицины, ветеринарии и биотехнологии. Биомедицина. 2011;(4):37-42. Tikhonov V.N., Bobovich V.E., Zaporozhets V.I. Creation of new generation of supersmall laboratory pigs for work in the field of medicine, veterinary science and biotechnology. Journal Biomed. 2011;(4):37-42. (In Russ.)
- Balzer D. Pulmonary valve replacement for tetralogy of Fallot. Methodist Debakey Cardiovasc J. 2019;15(2):122-132. PMID: 31384375; PMCID: PMC6668735. https://doi.org/10.14797/mdcj-15-2-122
- Stout K.K., Daniels C.J., Aboulhosn J.A., Bozkurt B., Broberg C.S., Colman J.M., Crumb S.R., Dearani J.A., Fuller S., Gurvitz M., Khairy P., Landzberg M.J., Saidi A., Valente A.M., Van Hare G.F. 2018 AHA/ACC Guideline for the management of adults with congenital heart disease: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73(12):1494-1563. PMID: 30121240. https://doi.org/10.1016/j.jacc.2018.08.1028
- Corno A.F. Pulmonary valve regurgitation: neither interventional nor surgery fits all. Front Pediatr. 2018;6:169. PMID: 29951475; PMCID: PMC6008531. https://doi.org/10.3389/fped.2018.00169
- Манукян С.Н., Сойнов И.А., Войтов А.В., Рзаева К.А., Баранов А.А., Богачев-Прокофьев А.В. Современные возможности транскатетерного протезирования клапана легочной артерии. Хирургия. Журнал им. Н.И. Пирогова. 2024;(2):32‑44. https://doi.org/10.17116/hirurgia202402132 Manukyan S.N., Soynov I.A., Voitov A.V., Rzaeva K.A., Baranov A.A., Bogachev-Prokofiev A.V. Modern possibilities for transcatheter pulmonary valve replacement. Pirogov Russian Journal of Surgery. 2024;(2):32‑44. (In Russ.) https://doi.org/10.17116/hirurgia202402132
- Kim A.Y., Jung J.W., Jung S.Y., Shin J.I., Eun L.Y., Kim N.K., Choi J.Y. Early outcomes of percutaneous pulmonary valve implantation with Pulsta and Melody valves: the first report from Korea. J Clin Med. 2020;9(9):2769. PMID: 32859019; PMCID: PMC7565703. https://doi.org/10.3390/jcm9092769
- Рзаева К.А., Тимченко Т.П., Журавлева И.Ю., Архипов А.Н., Горбатых А.В., Войтов А.В., Ничай Н.Р., Богачев-Прокофьев А.В., Сойнов И.А. Технические характеристики самораскрывающегося клапана для лечения клапанной патологии легочной артерии. Патология кровообращения и кардиохирургия. 2022;26(3):85-90. https://doi.org/10.21688/1681-3472-2022-3-85-90 Rzaeva K.A., Timchenko T.P., Zhuravleva I.Yu., Arkhipov A.N., Gorbatykh A.V., Voitov A.V., Nichay N.R., Bogachev-Prokophiev A.V., Soynov I.A. Technical features of a self-expandable prosthetic valve for the treatment of pulmonary valve disease. Patologiya krovoobrashcheniya i kardiokhirurgiya = Circulation Pathology and Cardiac Surgery. 2022;26(3):85-90. (In Russ.) https://doi.org/10.21688/1681-3472-2022-3-85-90
- Рзаева К.А., Тимченко Т.П., Журавлева И.Ю., Архипов А.Н., Горбатых А.В., Войтов А.В., Богачев-Прокофьев А.В., Сойнов И.А. Транскатетерная имплантация самораскрывающегося клапана легочной артерии в эксперименте на животных. Клиническая и экспериментальная хирургия. Журнал имени академика Б.В. Петровского. 2023;11(1):47-53. https://doi.org/10.33029/2308-1198-2023-11-1-47-53 Rzaeva K.A., Timchenko T.P., Zhuravleva I.Yu., Arkhipov A.N., Gorbatykh A.V., Voitov A.V., Bogachev-Prokophiev A.V., Soynov I.A. Transcatheter implantation of a self-expanding pulmonary valve in animal experiment. Clinical and Experimental Surgery. Petrovsky Journal. 2023;11(1):47-53. (In Russ.) https://doi.org/10.33029/2308-1198-2023-11-1-47-53
- Сойнов И.А., Манукян С.Н., Рзаева К.А., Войтов А.В., Тимченко Т.П., Кобелев Е., Архипов А.Н., Ничай Н.Р., Кулябин Ю.Ю., Журавлева И.Ю., Богачев-Прокофьев А.В. Варианты дисфункций пути оттока из правого желудочка. Кардиология и сердечно-сосудистая хирургия. 2023;16(4):351‑357. https://doi.org/10.17116/kardio202316041351 Soynov I.A., Manukyan S.N., Rzaeva K.A., Voitov A.V., Timchenko T.P., Kobelev E., Arkhipov A.N., Nichai N.R., Kulyabin Yu.Yu., Zhuravleva I.Yu., Bogachev-Prokofiev A.V. Dysfunctions of right ventricular outflow tract. Russian Journal of Cardiology and Cardiovascular Surgery. 2023;16(4):351‑357. (In Russ., In Engl.) https://doi.org/10.17116/kardio202316041351
- Сойнов И.А., Рзаева К.А., Горбатых А.В., Войтов А.В., Архипов А.Н., Ничай Н.Р., Кулябин Ю.Ю., Манукян С.Н., Богачев-Прокофьев А.В. Физико-механические свойства кондуитов при формировании пути оттока в легочную артерию. Комплексные проблемы сердечно-сосудистых заболеваний. 2024;13(1):67-76. https://doi.org/10.17802/2306-1278-2024-13-1-67-76 Soynov I.A., Rzaeva K.A., Gorbatykh A.V., Voitov A.V., Arkhipov A.N., Nichay N.R., Kulyabin Yu.Yu., Manukyan S.N., Bogachev-Prokophiev A.V. Physical and mechanical properties of conduits during the formation of the outflow tract into the pulmonary artery. Complex Issues of Cardiovascular Diseases. 2024;13(1):67-76. (In Russ.) https://doi.org/10.17802/2306-1278-2024-13-1-67-76
- Войтов А.В., Манукян С.Н., Владимиров С.В., Бородин В.П., Кобелев Е., Журавлева И.Ю., Архипов А.Н., Горбатых А.В., Рзаева К.А., Ничай Н.Р., Богачев-Прокофьев А.В., Сойнов И.А. Имплантация самораскрывающегося каркаса транскатетерного клапана in vitro в 3D модель сердца пациента с дисфункцией пути оттока из правого желудочка. Сибирский журнал клинической и экспериментальной медицины. 2024;39(2):104-111. https://doi.org/10.29001/2073-8552-2024-39-2-104-111 Voitov A.V., Manukian S.N., Vladimirov S.V., Borodin V.P., Kobelev E., Zhuravleva I.Yu., Arkhipov A.N., Gorbatykh A.V., Rzaeva K.A., Nichay N.R., Bogachev-Prokophiev A.V., Soynov I.A. Implantation of a self-expanding transcatheter valve in vitro into a 3D heart model of a patient with right ventricular outflow tract dysfunction. Siberian Journal of Clinical and Experimental Medicine. 2024;39(2):104-111. (In Russ.) https://doi.org/10.29001/2073-8552-2024-39-2-104-111
- Baessato F., Ewert P., Meierhofer C. CMR and percutaneous treatment of pulmonary regurgitation: outreach the search for the best candidate. Life (Basel). 2023;13(5):1127. PMID: 37240773; PMCID: PMC10222893. https://doi.org/10.3390/life13051127
- Horlick E.M., Haas N.A. Percutaneous pulmonary valve replacement: what a difference a day makes. J Am Coll Cardiol. 2020;76(24):2859-2861. PMID: 33303075. https://doi.org/10.1016/j.jacc.2020.11.003
- Nichay N.R., Zhuravleva I.Y., Kulyabin Y.Y., Zubritskiy A.V., Voitov A.V., Soynov I.A., Gorbatykh A.V., Bogachev-Prokophiev A.V., Karaskov A.M. Diepoxy- versus glutaraldehyde-treated xenografts: outcomes of right ventricular outflow tract reconstruction in children. World J Pediatr Congenit Heart Surg. 2020;11(1):56-64. PMID: 31835985. https://doi.org/10.1177/2150135119885900
- Nichay N.R., Dokuchaeva A.A., Kulyabin Yu.Yu., Boyarkin E.V., Kuznetsova E.V., Rusakova Ya.L., Murashov I.S., Vaver A.A., Bogachev-Prokophiev A.V., Zhuravleva I.Yu. Epoxy- versus glutaraldehyde-treated bovine jugular vein conduit for pulmonary valve replacement: a comparison of morphological changes in a pig model. Biomedicines. 2023;11(11):3101. PMID: 38002101; PMCID: PMC10669752. https://doi.org/10.3390/biomedicines11113101

