Prevention of respiratory complications in patients at risk of adverse respiratory events by perioperative nitric oxide delivery during cardiac surgery under cardiopulmonary bypass: a single-center prospective randomized study
Published 2024-11-08
Keywords
- Cardiac Surgical Procedures,
- Cardiopulmonary Bypass,
- Complications,
- Nitric Oxide,
- Oxygen
- Prospective Studies,
- Respiratory Failure ...More
How to Cite
Copyright (c) 2024 Kravchenko I.V., Podoksenov Yu.K., Tyo M.A., Kalashnikova T.P., Churilina E.A., Kozulin M.S., Gusakova A.M., Kozlov B.N., Kamenshchikov N.O.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Introduction: Organoprotective effects of perioperative nitric oxide delivery towards the heart and kidneys have been demonstrated many times in cardiac surgery but information about its ability to prevent respiratory complications is extremely limited.
Objective: This study aimed at evaluation of the efficacy and the safety of perioperative nitric oxide delivery for preventing respiratory complications during cardiac surgeries involving cardiopulmonary bypass in patients with an increased risk of adverse respiratory events and the identification of the risk factors towards postoperative acute respiratory.
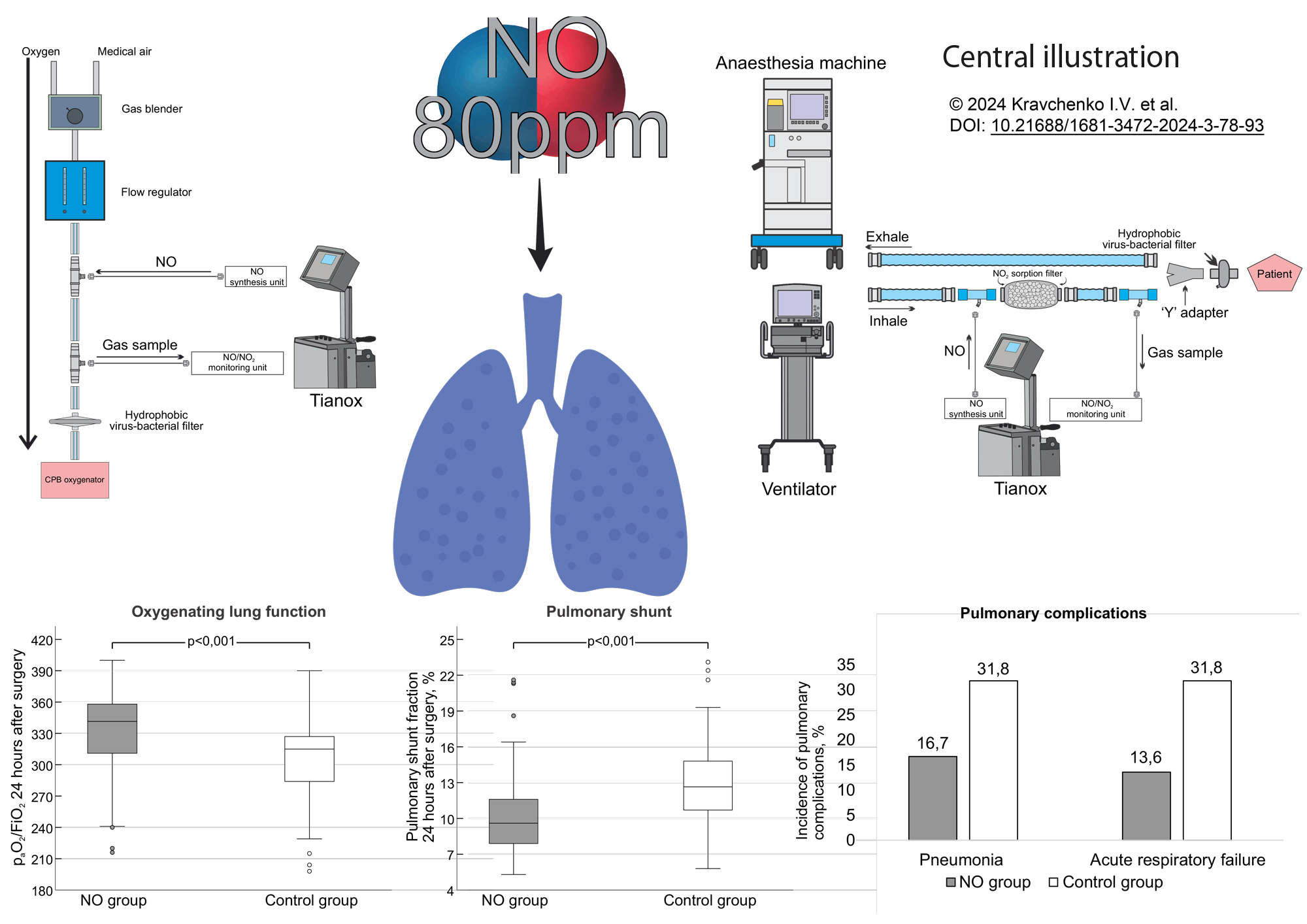
Methods: A prospective randomised controlled study included 132 cardiac surgical patients with increased risk of postoperative respiratory failure who were divided into two groups. The main group received nitric oxide at a concentration of 80 ppm throughout the operation and the first 6 hours at the postoperative period. The primary endpoint was the ratio of the partial pressure of oxygen in arterial blood to the fraction of inspired oxygen in 24 hours after the operation.
Results: Patients receiving perioperative nitric oxide demonstrated significant improvements in oxygenation (paO2/FiO2 ratio, p < 0.001) in 24 hour post-surgery period that was associated with a decrease in pulmonary shunt fraction (p < 0.001), a decline of acute respiratory failure incidence (p = 0.042), and postoperative pneumonia (p = 0.013). Delivery of nitric oxide did not entail an increase in methemoglobin concentration above 5% and nitrogen dioxide above 3 ppm. In 24 h after surgery, revealed cases of acute respiratory failure were associated with blood transfusion (p = 0.033), as well as an excess of cardiopulmonary bypass duration of more than 97 min and mechanical ventilator duration of more than 628 min.
Conclusion: Perioperative delivery of 80 ppm nitric oxide in cardiac surgeries involving cardiopulmonary bypass is safe and effective since it results in improved pulmonary oxygenating function and decreased pulmonary complications. In patients with an increased risk of adverse respiratory events, identified risk factors of acute respiratory failure include blood transfusion, prolonged cardiopulmonary bypass, and extended mechanical ventilation.
Received 23 September 2024. Revised 2 October 2024. Accepted 7 October 2024.
Funding
The study was carried out within the framework of the state assignment under the topic of basic scientific research No. 122123000017-3.
Conflict of interests
The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: N.O. Kamenshchikov, I.V. Kravchenko, Yu.K. Podoksenov, B.N. Kozlov
Data collection and analysis: I.V. Kravchenko, M.A. Tyo, E.A. Churilina, M.S. Kozulin, A.M. Gusakova, T.P. Kalashnikova
Statistical analysis: I.V. Kravchenko, Yu.K. Podoksenov
Drafting the article: I.V. Kravchenko, Yu.K. Podoksenov
Critical revision of the article: N.O. Kamenshchikov, B.N. Kozlov
Final approval of the version to be published: I.V. Kravchenko, Yu.K. Podoksenov, M.A. Tyo, T.P. Kalashnikova, E.A. Churilina, M.S. Kozulin, A.M. Gusakova, B.N. Kozlov, N.O. Kamenshchikov
References
- Badenes R., Lozano A., Belda F.J. Postoperative pulmonary dysfunction and mechanical ventilation in cardiac surgery. Crit Care Res Pract. 2015;2015:420513. PMID: 25705516; PMCID: PMC4332756. https://doi.org/10.1155/2015/420513
- Morariu A.M., Loef B.G., Aarts L.P., Rietman G.W., Rakhorst G., van Oeveren W., Epema A.H. Dexamethasone: benefit and prejudice for patients undergoing on-pump coronary artery bypass grafting: a study on myocardial, pulmonary, renal, intestinal, and hepatic injury. Chest. 2005;128(4):2677-2687. PMID: 16236942. https://doi.org/10.1378/chest.128.4.2677
- Еременко А.А., Зюляева Т.П. Проблема послеоперационной острой дыхательной недостаточности в кардиохирургии. Хирургия. Журнал им. Н.И. Пирогова. 2019;(8):5‑11. https://doi.org/10.17116/hirurgia20190815 Eremenko A.A., Zulyaeva T.P. Postoperative acute respiratory failure in cardiac surgery. Pirogov Russian Journal of Surgery. 2019;(8):5‑11. (In Russ.) https://doi.org/10.17116/hirurgia20190815
- Zainab A., Nguyen D.T., Graviss E.A., Fatima S., Masud F.N., MacGillivray T.E. Development and validation of a risk score for respiratory failure after cardiac surgery. Ann Thorac Surg. 2022;113(2):577-584. PMID: 33839130. https://doi.org/10.1016/j.athoracsur.2021.03.082
- Mohamed M.A., Cheng C., Wei X. Incidence of postoperative pulmonary complications in patients undergoing minimally invasive versus median sternotomy valve surgery: propensity score matching. J Cardiothorac Surg. 2021;16(1):287. PMID: 34627311; PMCID: PMC8501915. https://doi.org/10.1186/s13019-021-01669-7
- Baehner T., Boehm O., Probst C., Poetzsch B., Hoeft A., Baumgarten G., Knuefermann P. Kardiopulmonaler Bypass in der Herzchirurgie [Cardiopulmonary bypass in cardiac surgery]. Anaesthesist. 2012;61(10):846-856. PMID: 22971923. https://doi.org/10.1007/s00101-012-2050-0
- Apostolakis E., Filos K.S., Koletsis E., Dougenis D. Lung dysfunction following cardiopulmonary bypass. J Card Surg. 2010;25(1):47-55. PMID: 19549041. https://doi.org/10.1111/j.1540-8191.2009.00823.x
- Clark S.C. Lung injury after cardiopulmonary bypass. Perfusion. 2006;21(4):225-228. PMID: 16939116. https://doi.org/10.1191/0267659106pf872oa
- Bux J., Sachs U.J. The pathogenesis of transfusion-related acute lung injury (TRALI). Br J Haematol. 2007;136(6):788-799. PMID: 17341264. https://doi.org/10.1111/j.1365-2141.2007.06492.x
- Babik B., Asztalos T., Peták F., Deák Z.I., Hantos Z. Changes in respiratory mechanics during cardiac surgery. Anesth Analg. 2003;96(5):1280-1287. PMID: 12707120. https://doi.org/10.1213/01.ANE.0000055363.23715.40
- Matsugi E., Takashima S., Doteguchi S., Kobayashi T., Okayasu M. Real-world safety and effectiveness of inhaled nitric oxide therapy for pulmonary hypertension during the perioperative period of cardiac surgery: a post-marketing study of 2817 patients in Japan. Gen Thorac Cardiovasc Surg. 2024;72(5):311-323. PMID: 37713058; PMCID: PMC11018662. https://doi.org/10.1007/s11748-023-01971-2
- Muenster S., Zarragoikoetxea I., Moscatelli A., Balcells J., Gaudard P., Pouard P., Marczin N., Janssens S.P. Inhaled NO at a crossroads in cardiac surgery: current need to improve mechanistic understanding, clinical trial design and scientific evidence. Front Cardiovasc Med. 2024;11:1374635. PMID: 38646153; PMCID: PMC11027901. https://doi.org/10.3389/fcvm.2024.1374635
- Yan Y., Kamenshchikov N., Zheng Z., Lei C. Inhaled nitric oxide and postoperative outcomes in cardiac surgery with cardiopulmonary bypass: A systematic review and meta-analysis. Nitric Oxide. 2024;146:64-74. PMID: 38556145. https://doi.org/10.1016/j.niox.2024.03.004
- Chello M., Mastroroberto P., Perticone F., Celi V., Colonna A. Nitric oxide modulation of neutrophil-endothelium interaction: difference between arterial and venous coronary bypass grafts. J Am Coll Cardiol. 1998;31(4):823-826. PMID: 9525554. https://doi.org/10.1016/s0735-1097(97)00560-3
- Sawicki G., Salas E., Murat J., Miszta-Lane H., Radomski M.W. Release of gelatinase A during platelet activation mediates aggregation. Nature. 1997;386(6625):616-619. PMID: 9121586. https://doi.org/10.1038/386616a0
- Andrabi S.M., Sharma N.S., Karan A., Shahriar S.M.S., Cordon B., Ma B., Xie J. Nitric oxide: physiological functions, delivery, and biomedical applications. Adv Sci (Weinh). 2023;10(30):e2303259. PMID: 37632708; PMCID: PMC10602574. https://doi.org/10.1002/advs.202303259
- Van Dervort A.L., Yan L., Madara P.J., Cobb J.P., Wesley R.A., Corriveau C.C., Tropea M.M., Danner R.L. Nitric oxide regulates endotoxin-induced TNF-alpha production by human neutrophils. J Immunol. 1994;152(8):4102-4109. PMID: 8144975.
- Hataishi R., Rodrigues A.C., Neilan T.G., Morgan J.G., Buys E., Shiva S., Tambouret R., Jassal D.S., Raher M.J., Furutani E., Ichinose F., Gladwin M.T., Rosenzweig A., Zapol W.M., Picard M.H., Bloch K.D., Scherrer-Crosbie M. Inhaled nitric oxide decreases infarction size and improves left ventricular function in a murine model of myocardial ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol. 2006;291(1):H379-384. PMID: 16443673. https://doi.org/10.1152/ajpheart.01172.2005
- Kamenshchikov N.O., Safaee Fakhr B., Kravchenko I.V., Dish A.Y., Podoksenov Y.K., Kozlov B.N., Kalashnikova T.P., Tyo M.A., Anfinogenova N.D., Boshchenko A.A., Berra L. Assessment of continuous low-dose and high-dose burst of inhaled nitric oxide in spontaneously breathing COVID-19 patients: A randomized controlled trial. Nitric Oxide. 2024;149:41-48. PMID: 38880198. https://doi.org/10.1016/j.niox.2024.06.003
- Kamenshchikov N.O., Duong N., Berra L. Nitric oxide in cardiac surgery: a review article. Biomedicines. 2023;11(4):1085. PMID: 37189703; PMCID: PMC10135597. https://doi.org/10.3390/biomedicines11041085
- Пичугин В.В., Домнин С.Е., Калинина М.Л., Богуш А.В., Чигинев В.А., Гамзаев А.Б., Журко С.А. Интраоперационная NO терапия у пациентов с высокой легочной гипертензией при коррекции клапанных пороков сердца. Медицинский альманах. 2017;48(3):144-149. Pichugin V.V., Domnin S.E., Kalinina M.L., Bogush A.V., Chiginev V.A., Gamzaev A.B., Zhurko S.A. Intraoperative NO therapy in patients with high pulmonary hypertension during correction of valve heart defects. Medicinskij almanah = Medical Almanac. 2017;48(3):144-149. (In Russ.)
- Jammer I., Wickboldt N., Sander M., Smith A., Schultz M.J., Pelosi P., Leva B., Rhodes A., Hoeft A., Walder B., Chew M.S., Pearse R.M., European Society of Anaesthesiology (ESA) and the European Society of Intensive Care Medicine (ESICM). Standards for definitions and use of outcome measures for clinical effectiveness research in perioperative medicine: European Perioperative Clinical Outcome (EPCO) definitions: a statement from the ESA-ESICM joint taskforce on perioperative outcome measures. Eur J Anaesthesiol. 2015;32(2):88-105. PMID: 25058504. https://doi.org/10.1097/EJA.0000000000000118
- Заболотских И.Б., Грицан А.И., Киров М.Ю., Кузовлев А.Н., Лебединский К.М., Мазурок В.А., Проценко Д.Н., Трембач Н.В., Шадрин Р.В., Ярошецкий А.И. Периоперационное ведение пациентов с дыхательной недостаточностью: методические рекомендации Общероссийской общественной организации «Федерация анестезиологов и реаниматологов». Вестник интенсивной терапии им. А.И. Салтанова. 2022;(4):7-23. https://doi.org/10.21320/1818-474X-2022-4-7-23 Zabolotskikh I.B., Gritsan A.I., Kirov M.Yu., Kuzovlev A.N., Lebedinskii K.M., Mazurok V.A., Protsenko D.N., Trembach N.V., Shadrin R.V., Yaroshetskiy A.I. Perioperative management of patients with respiratory failure: methodological recommendations of the All-Russian public organization “Federation of Anesthesiologists and Reanimatologists.” Annals of Critical Care. 2022;(4):7-23. (In Russ.) https://doi.org/10.21320/1818-474X-2022-4-7-23
- Кравченко И.В., Геренг Е.А., Подоксенов Ю.К., Тё М.А., Серебрякова О.Н., Бянкина М.А., Горохова А.В., Козлов Б.Н., Мильто И.В., Каменщиков Н.О. Влияние доставки оксида азота на морфофункциональное состояние легких при моделировании искусственного кровообращения: экспериментальное исследование. Пульмонология. 2024;34(3):385-394. https://doi.org/10.18093/0869-0189-2024-34-3-385-394 Kravchenko I.V., Gereng E.A., Podoksenov Yu.K., Tyo M.A., Serebryakova O.N., Byankina M.A., Gorokhova A.V., Kozlov B.N., Milto I.V., Kamenshchikov N.O. Effect of nitric oxide supply on the morphofunctional state of the lungs during cardiopulmonary bypass modelling: an experimental study. Pulmonologiya. 2024;34(3):385-394. (In Russ.) https://doi.org/10.18093/0869-0189-2024-34-3-385-394
- Kamenshchikov N.O., Diakova M.L., Podoksenov Yu.K., Churilina E.A., Rebrova T.Yu., Akhmedov S.D., Maslov L.N., Mukhomedzyanov A.V., Kim E.B., Tokareva E.S., Kravchenko I.V., Boiko A.M., Kozulin M.S., Kozlov B.N. Potential mechanisms for organoprotective effects of exogenous nitric oxide in an experimental study. Biomedicines. 2024;12(4):719. PMID: 38672075; PMCID: PMC11048067. https://doi.org/10.3390/biomedicines12040719
- Engels G.E., van Oeveren W. Biomarkers of lung injury in cardiothoracic surgery. Dis Markers. 2015;2015:472360. PMID: 25866435; PMCID: PMC4381722. https://doi.org/10.1155/2015/472360
- Wang D., Huang X., Wang H., Le S., Yang H., Wang F., Du X. Risk factors for postoperative pneumonia after cardiac surgery: a prediction model. J Thorac Dis. 2021;13(4):2351-2362. PMID: 34012584; PMCID: PMC8107540. https://doi.org/10.21037/jtd-20-3586
- Deppisch C., Herrmann G., Graepler-Mainka U., Wirtz H., Heyder S., Engel C., Marschal M., Miller C.C., Riethmüller J. Gaseous nitric oxide to treat antibiotic resistant bacterial and fungal lung infections in patients with cystic fibrosis: a phase I clinical study. Infection. 2016;44(4):513-520. PMID: 26861246. https://doi.org/10.1007/s15010-016-0879-x
- Ghaffari A., Miller C.C., McMullin B., Ghahary A. Potential application of gaseous nitric oxide as a topical antimicrobial agent. Nitric Oxide. 2006;14(1):21-29. PMID: 16188471. https://doi.org/10.1016/j.niox.2005.08.003
- Miller C.C., Rawat M., Johnson T., Av-Gay Y. Innate protection of Mycobacterium smegmatis against the antimicrobial activity of nitric oxide is provided by mycothiol. Antimicrob Agents Chemother. 2007;51(9):3364-3366. PMID: 17638697; PMCID: PMC2043170. https://doi.org/10.1128/AAC.00347-07
- Miller C., McMullin B., Ghaffari A., Stenzler A., Pick N., Roscoe D., Ghahary A., Road J., Av-Gay Y. Gaseous nitric oxide bactericidal activity retained during intermittent high-dose short duration exposure. Nitric Oxide. 2009;20(1):16-23. PMID: 18789393. https://doi.org/10.1016/j.niox.2008.08.002
- Thanavaro J., Taylor J., Vitt L., Guignon M.S., Thanavaro S. Predictors and outcomes of postoperative respiratory failure after cardiac surgery. J Eval Clin Pract. 2020;26(5):1490-1497. PMID: 31876045. https://doi.org/10.1111/jep.13334
- Кузьков В.В., Лапин К.С., Фот Е.В., Киров М.Ю. Вентилятор-ассоциированное повреждение легких в отделении интенсивной терапии и операционной — что нового? Вестник анестезиологии и реаниматологии. 2020;17(5):47-61. https://doi.org/10.21292/2078-5658-2020-17-5-47-61 Kuzkov V.V., Lapin K.S., Fot E.V., Kirov M.Yu. Ventilator-associated lung injury in the intensive care unit and operating room — what's new? Messenger of Anesthesiology and Resuscitation. 2020;17(5):47-61. (In Russ.) https://doi.org/10.21292/2078-5658-2020-17-5-47-61

