Published 2023-09-29
Keywords
- Cardiopulmonary Bypass,
- Comorbidity,
- Extracorporeal Circulation,
- Retrospective Studies,
- Triggering Receptor Expressed on Myeloid Cells-1
How to Cite
Copyright (c) 2023 Revishvili A.Sh., Kornelyuk R.A., Plotnikov G.P., Berikashvili L.B., Komkov I.P., Malyshenko E.S., Zemskov V.M., Popov V.A.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Background: As the severity of comorbid diseases increases, risks of cardiopulmonary bypass (CPB) complications significantly increase. A complex of the procedure’s damaging factors provokes a systemic inflammatory response that in some cases is accompanied by the damage to target organs, transitioning from chronic organ dysfunctions into acute ones. Some studies on the use of minimal invasive extracorporeal circulation (MiECC) systems show their advantage over conventional extracorporeal circulation (CECC); however, the diversity of MiECC systems, patients, and outcomes precludes us from confidently extrapolating these data to older comorbid patients.
Objective: To compare the severity of systemic inflammation and treatment outcomes in comorbid patients who underwent cardiac surgery with CECC and MiECC.
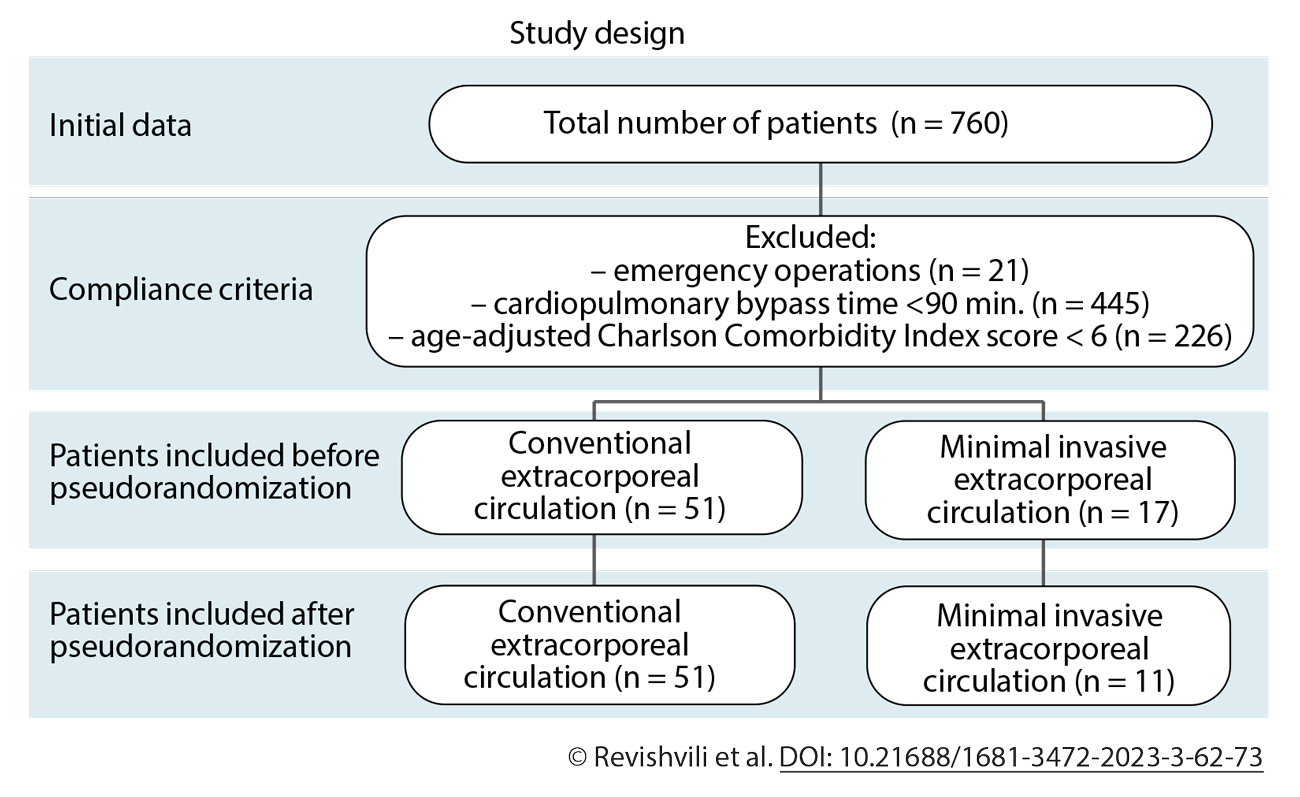
Methods: We conducted a retrospective cohort study of 760 patients who consecutively underwent elective cardiac operations in 2019-2022. Inclusion criteria: comorbid status (age-adjusted Charlson Comorbidity Index score of ≥6); CPB time ≥90 min. Exclusion criteria: emergency surgery, refusal to participate in the study. A total of 68 patients met the inclusion criteria. We formed 2 study groups based on the extracorporeal circulation method: CECC group (n = 51) and MiECC group (n = 17). Control points: before CPB and 24 hours after the surgery (lactate; creatinine; oxygenation index, hemolysis level). For systemic inflammatory response markers: 1 hour after the CPB start and 24 hours after the CPB end (interleukin 6; interleukin 10; procalcitonin; C-reactive protein; soluble Triggering Receptor Expressed on Myeloid Cells-1 [sTREM-1]). We evaluated respiratory and renal complications, drainage-related hemorrhages, hemostatic disorders, the need for sympathomimetic drugs, and the length of stay in an intensive care unit and inpatient hospital.
Results: Between the groups there were no statistically significant differences in gender and anthropometric characteristics, surgery types, and main perfusion parameters. In the CECC group, we observed significantly higher doses of vasoconstrictors (norepinephrine) as well as a decrease in urine output and lung injury and an increase in lactate and hemolysis. The systemic inflammatory response markers were also significantly higher.
Conclusion: Compared with CECC, MiECC does not significantly affect the frequency of organ dysfunctions; however, it reduces the severity of the systemic inflammatory response and immune suppression that are trigger mechanisms for multiple organ dysfunction syndrome. It is particularly important for patients with chronic organ dysfunctions. A range of indications for MiECC systems should be defined given its high cost and off-target effect on pleiotropic factors of systemic inflammatory response development.
Received 6 April 2023. Revised 17 August 2023. Accepted 18 August 2023.
Funding: The study was conducted within the framework of the research project "Development of minimally invasive and hybrid technologies for surgical treatment of heart diseases".
Conflict of interest: The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: A.Sh. Revishvili, G.P. Plotnikov, V.A. Popov
Data collection and analysis: R.A. Kornelyuk, L.B. Berikashvili, I.P. Komkov, E.S. Malyshenko, V.M. Zemskov
Statistical analysis: L.B. Berikashvili
Drafting the article: R.A. Kornelyuk, G.P. Plotnikov, L.B. Berikashvili
Critical revision of the article: G.P. Plotnikov
Final approval of the version to be published: A.Sh. Revishvili, R.A. Kornelyuk, G.P. Plotnikov, L.B. Berikashvili, I.P. Komkov, E.S. Malyshenko, V.M. Zemskov, V.A. Popov
References
- Anastasiadis K., Argiriadou H., Deliopoulos A., Antonitsis P. Minimal invasive extracorporeal circulation (MiECC): the state-of-the-art in perfusion. J Thorac Dis. 2019;11(Suppl 10):S1507-S1514. PMID: 31293801; PMCID: PMC6586580. https://doi.org/10.21037/jtd.2019.01.66
- Соколова Н.Ю., Голухова Е.З. Коморбидность в прогнозировании отдаленных результатов реваскуляризации миокарда у больных ишемической болезнью сердца. Кардиология и сердечно-сосудистая хирургия. 2018;11(5):22-27. https://doi.org/10.17116/kardio20181105122 Sokolova N.Yu., Golukhova E.Z. Comorbidity in predicting the long-term outcomes of myocardial revascularization in patients with coronary artery disease. Kardiologiya i serdechno-sosudistaya khirurgiya = Russian Journal of Cardiology and Cardiovascular Surgery. 2018;11(5):22-27. (In Russ.) https://doi.org/10.17116/kardio20181105122
- Барбараш О.Л., Семенов В.Ю., Самородская И.В., Евсеева М.В., Рожков Н.А., Сумин А.Н., Барбараш Л.С. Коморбидная патология у больных ишемической болезнью сердца при коронарном шунтировании: опыт двух кардиохирургических центров. Российский кардиологический журнал. 2017;(3):6-13. https://doi.org/10.15829/1560-4071-2017-3-6-13 Barbarash O.L., Semjonov V.Yu., Samorodskaya I.V., Evseeva М.V., Rozhkov N.A., Sumin A.N., Barbarash L.S. Comorbidity in coronary heart disease patients undergoing bypass grafting: an experience of two surgery centers. Russian Journal of Cardiology. 2017;(3):6-13. (In Russ.) https://doi.org/10.15829/1560-4071-2017-3-6-13
- Yellon D.M., Hausenloy D.J. Myocardial reperfusion injury. N Engl J Med. 2007;357(11):1121-1135. PMID: 17855673. https://doi.org/10.1056/NEJMra071667
- Day J.R.S., Taylor K.M. The systemic inflammatory response syndrome and cardiopulmonary bypass. Int J Surg. 2005;3(2):129-140. PMID: 17462274. https://doi.org/10.1016/j.ijsu.2005.04.002
- Angelini G.D., Reeves B.C., Evans J., COMICS investigators. Conventional versus minimally invasive extracorporeal circulation in patients undergoing cardiac surgery: protocol for a randomised controlled trial (COMICS). Perfusion. 2021;36(4):388-394. PMID: 32781894; PMCID: PMC8167910. https://doi.org/10.1177/0267659120946731
- Anastasiadis K., Murkin J., Antonitsis P., Bauer A., Ranucci M., Gygax E., Schaarschmidt J., Fromes Y., Philipp A., Eberle B., Punjabi P., Argiriadou H., Kadner A., Jenni H., Albrecht G., van Boven W., Liebold A., de Somer F., Hausmann H., Deliopoulos A., El-Essawi A., Mazzei V., Biancari F., Fernandez A., Weerwind P., Puehler T., Serrick C., Waanders F., Gunaydin S., Ohri S., Gummert J., Angelini G., Falk V., Carrel T. Use of minimal invasive extracorporeal circulation in cardiac surgery: principles, definitions and potential benefits. A position paper from the Minimal invasive Extra-Corporeal Technologies international Society (MiECTiS). Interact Cardiovasc Thorac Surg. 2016;22(5):647-662. PMID: 26819269; PMCID: PMC4892134. https://doi.org/10.1093/icvts/ivv380
- Biancari F., Rimpiläinen R. Meta-analysis of randomised trials comparing the effectiveness of miniaturised versus conventional cardiopulmonary bypass in adult cardiac surgery. Heart. 2009;95(12):964-969. PMID: 19342377. https://doi.org/10.1136/hrt.2008.158709
- Benedetto U., Ng C., Frati G., Biondi-Zoccai G., Vitulli P., Zeinah M., Raja S.G.; Cardiac Outcomes METa-analysis (COMET) group. Miniaturized extracorporeal circulation versus off-pump coronary artery bypass grafting: a meta-analysis of randomized controlled trials. Int J Surg. 2015;14:96-104. PMID: 25560750. https://doi.org/10.1016/j.ijsu.2014.12.021
- Kowalewski M., Pawliszak W., Kołodziejczak M., Navarese E.P., Anisimowicz L. 30-day mortality reduction with miniaturized extracorporeal circulation as compared to conventional cardiopulmonary bypass for coronary revascularization. Meta-analysis of randomized controlled trials. Int J Cardiol. 2015;198:63-65. PMID: 26151717. https://doi.org/10.1016/j.ijcard.2015.06.153
- Charlson M.E., Pompei P., Ales K.L., MacKenzie C.R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-383. PMID: 3558716. https://doi.org/10.1016/0021-9681(87)90171-8
- Charlson M., Szatrowski T.P., Peterson J., Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245-1251. PMID: 7722560. https://doi.org/10.1016/0895-4356(94)90129-5
- Belov A., Katkov K., Vinokurov I.A., Stonogin K., Komarov D. Cardiopulmonary bypass duration as predictor of immediate results after cardiac surgery. Khirurgiia (Mosk). 2015;(5):4-13. (Engl., Russ.) https://doi.org/10.17116/hirurgia201554-13
- Salis S., Mazzanti V.V., Merli G., Salvi L., Tedesco C.C., Veglia F., Sisillo E. Cardiopulmonary bypass duration is an independent predictor of morbidity and mortality after cardiac surgery. J Cardiothorac Vasc Anesth. 2008;22(6):814-822. PMID: 18948034. https://doi.org/10.1053/j.jvca.2008.08.004
- Adamik B., Kübler A., Gozdzik A., Gozdzik W. Prolonged cardiopulmonary bypass is a risk factor for intestinal ischaemic damage and endotoxaemia. Heart Lung Circ. 2017;26(7):717-723. PMID: 27956161. https://doi.org/10.1016/j.hlc.2016.10.012
- Clough R.A., Leavitt B.J., Morton J.R., Plume S.K., Hernandez F., Nugent W., Lahey S.J., Ross C.S., O'Connor G.T. The effect of comorbid illness on mortality outcomes in cardiac surgery. Arch Surg. 2002;137(4):428-432; discussion 432-433. PMID: 11926947. https://doi.org/10.1001/archsurg.137.4.428
- Scrutinio D., Giannuzzi P. Comorbidity in patients undergoing coronary artery bypass graft surgery: impact on outcome and implications for cardiac rehabilitation. Eur J Cardiovasc Prev Rehabil. 2008;15(4):379-385. PMID: 18677160. https://doi.org/10.1097/HJR.0b013e3282fd5c6f
- Mullan C.W., Mori M., Pichert M.D., Bin Mahmood S.U., Yousef S., Geirsson A. United States national trends in comorbidity and outcomes of adult cardiac surgery patients. J Card Surg. 2020;35(9):2248-2253. PMID: 33448476. https://doi.org/10.1111/jocs.14764
- Dessai S.B., Fasal R., Dipin J., Adarsh D., Balasubramanian S. Age-adjusted Charlson comorbidity index and 30-day morbidity in pelvic surgeries. South Asian J Cancer. 2018;7(4):240-243. PMID: 30430092; PMCID: PMC6190402. https://doi.org/10.4103/sajc.sajc_241_17
- Qu W.-F., Zhou P.-Y., Liu W.-R., Tian M.-X., Jin L., Jiang X.-F., Wang H., Tao C.-Y., Fang Y., Zhou Y.-F., Song S.-S., Ding Z.-B., Peng Y.-F., Dai Z., Qiu S.-J., Zhou J., Fan J., Tang Z., Shi Y.-H. Age-adjusted Charlson Comorbidity Index predicts survival in intrahepatic cholangiocarcinoma patients after curative resection. Ann Transl Med. 2020;8(7):487. PMID: 32395531; PMCID: PMC7210176. https://doi.org/10.21037/atm.2020.03.23
- Minol J.-P., Dimitrova V., Petrov G., Langner R., Boeken U., Rellecke P., Aubin H., Kamiya H., Sixt S., Huhn R., Sugimura Y., Albert A., Lichtenberg A., Akhyari P. The age-adjusted Charlson comorbidity index in minimally invasive mitral valve surgery. Eur J Cardiothorac Surg. 2019;56(6):1124-1130. PMID: 31501891. https://doi.org/10.1093/ejcts/ezz240
- Coyan G.N., Chin H., Shah A., Miguelino A.M., Wang Y., Kilic A., Sultan I., Sciortino C.M., Chu D. Charlson comorbidity index is associated with longer-term mortality and re-admissions following coronary artery bypass grafting. J Surg Res. 2022;275:300-307. PMID: 35313139. https://doi.org/10.1016/j.jss.2022.02.012
- Argiriadou H., Antonitsis P., Gkiouliava A., Papapostolou E., Deliopoulos A., Anastasiadis K. Minimal invasive extracorporeal circulation preserves coagulation integrity. Perfusion. 2022;37(3):257-265. PMID: 33637025. https://doi.org/10.1177/0267659121998544
- Modrau I.S., Halle D.R., Nielsen P.H., Kimose H.H., Greisen J.R., Kremke M., Hvas A.-M. Impact of minimally invasive extracorporeal circulation on coagulation — a randomized trial. Eur J Cardiothorac Surg. 2020;57(6):1145-1153. PMID: 32011717; PMCID: PMC7239600. https://doi.org/10.1093/ejcts/ezaa010
- Anastasiadis K., Asteriou C., Antonitsis P., Argiriadou H., Grosomanidis V., Kyparissa M., Deliopoulos A., Konstantinou D., Tossios P. Enhanced recovery after elective coronary revascularization surgery with minimal versus conventional extracorporeal circulation: a prospective randomized study. J Cardiothorac Vasc Anesth. 2013;27(5):859-864. PMID: 23791499. https://doi.org/10.1053/j.jvca.2013.01.010
- Bauer A., Diez C., Schubel J., El-Shouki N., Metz D., Eberle T., Hausmann H. Evaluation of hemodynamic and regional tissue perfusion effects of minimized extracorporeal circulation (MECC). J Extra Corpor Technol. 2010;42(1):30-39. PMID: 20437789; PMCID: PMC4680062.
- Huybregts R.A.J.M., Morariu A.M., Rakhorst G., Spiegelenberg S.R., Romijn H.W.A., de Vroege R., van Oeveren W. Attenuated renal and intestinal injury after use of a mini-cardiopulmonary bypass system. Ann Thorac Surg. 2007;83(5):1760-1766. PMID: 17462395. https://doi.org/10.1016/j.athoracsur.2007.02.016
- Immer F.F., Pirovino C., Gygax E., Englberger L., Tevaearai H., Carrel T.P. Minimal versus conventional cardiopulmonary bypass: assessment of intraoperative myocardial damage in coronary bypass surgery. Eur J Cardiothorac Surg. 2005;28(5):701-704. PMID: 16221550. https://doi.org/10.1016/j.ejcts.2005.08.019
- Григорьев Е.В., Шукевич Д.Л., Матвеева В.Г., Пугачев С.В., Каменева Е.А., Корнелюк Р.А. Миелоидные супрессорные клетки в патогенезе критических состояний. Патология кровообращения и кардиохирургия. 2016;20(3):20-25. https://doi.org/10.21688/1681-3472-2016-3-20-25 Grigoryev E.V., Shukevich D.L., Matveeva V.G., Pugachev S.V., Kameneva E.A., Kornelyuk R.A. Myeloid suppressor cells in the pathogenesis of critical states. Patologiya krovoobrashcheniya i kardiokhirurgiya = Circulation Pathology and Cardiac Surgery. 2016;20(3):20-25. (In Russ.) https://doi.org/10.21688-1681-3472-2016-3-20-25
- Матвеева В.Г., Ханова М.Ю., Ивкин А.А., Корнелюк Р.А., Григорьев Е.В. Иммуносупрессорный профиль пациентов, оперированных по поводу приобретенных пороков сердца в условиях искусственного кровообращения. Вестник интенсивной терапии имени А.И. Салтанова. 2020;(3):74-87. https://doi.org/10.21320/1818-474X-2020-3-74-87 Matveeva V.G., Khanova M.Yu., Ivkin A.A., Kornelyuk R.A., Grigoryev E.V. Immunosuppressive profile of patients operated for acquired heart diseases under artificial circulation. A prospective study. Annals of Critical Care. 2020;(3):74-87. (In Russ.) https://doi.org/10.21320/1818-474X-2020-3-74-87

