Structure and properties of fibrin/polyvinyl alcohol interpenetrating polymer network hydrogel as a modifying coating for small-diameter vascular grafts
Published 2023-06-30
Keywords
- Fibrin,
- Hydrogels,
- Polymerization,
- Polymers,
- Polyvinyl
- Tissue Engineering ...More
How to Cite
Copyright (c) 2023 Matveeva V.G., Rezvova M.A., Glushkova T.V., Sergeeva A.V., Krivkina E.O., Antonova L.V., Barbarash L.S.

This work is licensed under a Creative Commons Attribution 4.0 International License.
Abstract
Background: Interpenetrating polymer network (IPN) hybrid hydrogels enable regulating their properties by varying the composition and concentration of their components. Fibrin is an available natural polymer with ideal biological properties but low strength and tendency to retraction. Polyvinyl alcohol hydrogels are stable, comparable in strength to biological tissues but bioinert. Fibrin/polyvinyl alcohol (F/PVA) IPN can overcome the shortcomings of each component and create an improved material for tissue engineering.
Objective: To assess the possibility of and conditions for obtaining a homogeneous IPN by subsequent fibrin polymerization and polyvinyl alcohol cryostructuring while preserving as much as possible mechanical and biological properties beneficial for tissue engineering.
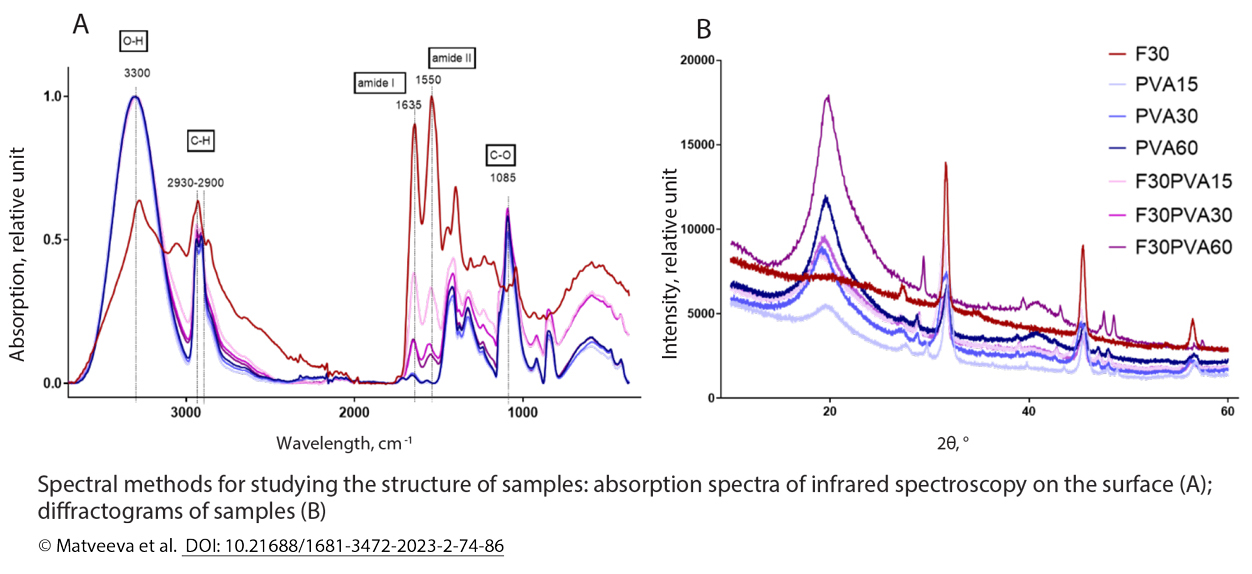
Methods: F/PVA IPN was obtained by subsequent fibrin polymerization (30 mg/mL) and polyvinyl alcohol cryostructuring (15, 30, and 60 mg/mL). We studied the structure using a scanning electron microscope, histology, infrared spectroscopy, and X-ray diffraction analysis. We tested mechanical properties and shrinkage of the samples. Biological features were assessed in vitro in terms of viability, cell count, proliferative and metabolic activity of EA.hy926 endothelial cell culture.
Results: Our study found the maximum amount of fibrin on the surface of F30PVA15 IPN and its minimum amount on the surface of F30PVA60. These results were supported by the high biological appeal of F30PVA15 compared with F30PVA30 and F30PVA60. F30PVA60 hydrogels demonstrated shrinkage resistance compared to the template; F30PVA30 and F30PVA15 samples decreased by 1.4 and 2.5 times, respectively. Although the mechanical strength of all monocomponent hydrogels and IPN samples did not compare to that of the internal thoracic vein, F30PVA30 and F30PVA60 demonstrated better results than F30PVA15 and fibrin alone.
Conclusion: Our method allows obtaining shrink-resistant IPN hydrogels with improved mechanical and tolerable biological properties at polyvinyl alcohol concentrations of > 15 mg/mL and < 60 mg/mL. However, the insufficient strength of this material limits its use in vascular engineering to a modifying coating.
Received 25 January 2023. Revised 17 March 2023. Accepted 29 March 2023.
Funding: This research was funded by the complex program of basic research under the Siberian Branch of the Russian Academy of Sciences within the basic research topic of Research Institute for Complex Issues of Cardiovascular Diseases No. 0419-2022-0001 “Molecular, cellular and biomechanical mechanisms of the pathogenesis of cardiovascular diseases in the development of new treatment methods based on personalized pharmacotherapy, minimally invasive medical devices, biomaterials and tissue-engineered implants”.
Conflict of interest: The authors declare no conflict of interest.
Contribution of the authors
Conception and study design: V.G. Matveeva, M.A. Rezvova
Data collection and analysis: V.G. Matveeva, M.A. Rezvova, T.V. Glushkova, E.O. Krivkina, A.V. Sergeeva
Statistical analysis: V.G. Matveeva, M.A. Rezvova, T.V. Glushkova, A.V. Sergeeva
Drafting the article: V.G. Matveeva
Critical revision of the article: V.G. Matveeva, M.A. Rezvova, L.V. Antonova, L.S. Barbarash
Final approval of the version to be published: V.G. Matveeva, M.A. Rezvova, T.V. Glushkova, A.V. Sergeeva, E.O. Krivkina, L.V. Antonova, L.S. Barbarash
References
- Mallis P., Kostakis A., Stavropoulos-Giokas C., Michalopoulos E. Future perspectives in small-diameter vascular graft engineering. Bioengineering (Basel). 2020;7(4):160. PMID: 33321830; PMCID: PMC7763104. https://doi.org/10.3390/bioengineering7040160
- Zizhou R., Wang X., Houshyar S. Review of polymeric biomimetic small-diameter vascular grafts to tackle intimal hyperplasia. ACS Omega. 2022;7(26):22125-22148. PMID: 35811906; PMCID: PMC9260943. https://doi.org/10.1021/acsomega.2c01740
- Chlupáč J., Filová E., Riedel T., Houska M., Brynda E., Remy-Zolghadri M., Bareille R., Fernandez P., Daculsi R., Bourget C., Bordenave L., Bačáková L. Attachment of human endothelial cells to polyester vascular grafts: pre-coating with adhesive protein assemblies and resistance to short-term shear stress. Physiol Res. 2014;63(2):167-177. PMID: 24397801. https://doi.org/10.33549/physiolres.932577
- Morin K.T., Tranquillo R.T. In vitro models of angiogenesis and vasculogenesis in fibrin gel. Exp Cell Res. 2013;319(16):2409-2417. PMID: 23800466; PMCID: PMC3919069. https://doi.org/10.1016/j.yexcr.2013.06.006
- Матвеева В.Г., Сенокосова Е.А., Ханова М.Ю., Глушкова Т.В., Антонова Л.В. Влияние способа полимеризации на свойства фибриновых матриц (пилотное исследование in vitro). Комплексные проблемы сердечно-сосудистых заболеваний. 2022;11(4S):134-145. https://doi.org/10.17802/2306-1278-2022-11-4S-134-145 Matveeva V.G., Senokosova E.A., Khanova M.Yu., Glushkova T.V., Antonova L.V. Influence of the polymerization method on the properties of fibrin matrices. Complex Issues of Cardiovascular Diseases. 2022;11(4S):134-145. (In Russ.) https://doi.org/10.17802/2306-1278-2022-11-4S-134-145
- Матвеева В.Г., Ханова М.Ю., Антонова Л.В., Барбараш Л.С. Фибрин — перспективный материал для тканевой сосудистой инженерии. Вестник трансплантологии и искусственных органов. 2020;22(1):196-208. https://doi.org/10.15825/1995-1191-2020-1-196-208 Matveeva V.G., Khanova M.U., Antonova L.V., Barbarash L.S. Fibrin — a promising material for vascular tissue engineering. Russian Journal of Transplantology and Artificial Organs. 2020;22(1):196-208. (In Russ.) https://doi.org/10.15825/1995-1191-2020-1-196-208
- Mironi-Harpaz I., Zigerson S., Seliktar D. Fibrin-based hydrogel scaffolds for controlling cell-matrix interaction in vascular tissue engineering. Advanced Biomaterials and Devices in Medicine. 2014;1(1):28-37. https://doi.org/10.24411/2409-2568-2014-00004
- Akpalo E., Bidault L., Boissière M., Vancaeyzeele C., Fichet O., Laretta-Garde V. Fibrin–polyethylene oxide interpenetrating polymer networks: new self-supported biomaterials combining the properties of both protein gel and synthetic polymer. Acta Biomater. 2011;7(6):2418-2427. PMID: 21382527. https://doi.org/10.1016/j.actbio.2011.03.002
- Deneufchâtel M., Larreta-Garde V., Fichet O. Polyethylene glycol-albumin/fibrin interpenetrating polymer networks with adaptable enzymatic degradation for tissue engineering applications. Polym Degrad Stab. 2018;152:218-227. https://doi.org/10.1016/j.polymdegradstab.2018.04.023
- Gsib O., Duval J.-L., Goczkowski M., Deneufchatel M., Fichet O., Larreta-Garde V., Bencherif A.S., Egles C. Evaluation of fibrin-based interpenetrating polymer networks as potential biomaterials for tissue engineering. Nanomaterials. 2017;7(12):436. PMID: 29232876; PMCID: PMC5746926. https://doi.org/10.3390/nano7120436
- Bidault L., Deneufchatel M., Vancaeyzeele C., Fichet O., Larreta-Garde V. Self-supported fibrin-polyvinyl alcohol interpenetrating polymer networks: an easily handled and rehydratable biomaterial. Biomacromolecules. 2013;14(11):3870-3879. PMID: 24050436. https://doi.org/10.1021/bm400991k
- Bidault L., Deneufchatel M., Hindié M., Vancaeyzeele C., Fichet O., Larreta-Garde V. Fibrin-based interpenetrating polymer network biomaterials with tunable biodegradability. Polymer. 2015;62:19-27. https://doi.org/10.1016/j.polymer.2015.02.014
- Ma S., Wang S., Li Q., Leng Y., Wang L., Hu G.-H. A novel method for preparing poly(vinyl alcohol) hydrogels: preparation, characterization, and application. Ind Eng Chem Res. 2017;56(28):7971-7976. https://doi.org/10.1021/acs.iecr.7b01812
- Stauffer S.R., Peppast N.A. Poly(vinyl alcohol) hydrogels prepared by freezing-thawing cyclic processing. Polymer. 1992;33(18):3932-3936. https://doi.org/10.1016/0032-3861(92)90385-A
- Shipp D.A. Reversible-deactivation radical polymerizations. Polym Rev. 2011;51(2):99-103. https://doi.org/10.1080/15583724.2011.566406
- Millon L.E., Nieh M.-P., Hutter J.L., Wan W.K. SANS Characterization of an anisotropic poly(vinyl alcohol) hydrogel with vascular applications. Macromolecules. 2007;40(10):3655-3662. https://doi.org/10.1021/ma062781f
- Kumar A., Han S.S. PVA-based hydrogels for tissue engineering: A review. Int J Polym Mater Polym Biomater. 2017;66(4):159-182. https://doi.org/10.1080/00914037.2016.1190930
- Jiang S., Liu S., Feng W. PVA hydrogel properties for biomedical application. J Mech Behav Biomed Mater. 2011;4(7):1228-1233. PMID: 21783131. https://doi.org/10.1016/j.jmbbm.2011.04.005
- Jiang Y., Schädlich A., Amado E., Weis C., Odermatt E., Mäder K., Kressler J. In vivo studies on intraperitoneally administrated poly(vinyl alcohol). J Biomed Mater Res B Appl Biomater. 2010;93(1):275-284. PMID: 20119945. https://doi.org/10.1002/jbm.b.31585
- Wang B.H., Campbell G. Formulations of polyvinyl alcohol cryogel that mimic the biomechanical properties of soft tissues in the natural lumbar intervertebral disc. Spine (Phila Pa 1976). 2009;34(25):2745-2753. PMID: 19940732. https://doi.org/10.1097/BRS.0b013e3181b4abf5
- Bourke S.L., Al-Khalili M., Briggs T., Michniak B.B., Kohn J., Poole-Warren L.A. A photo-crosslinked poly(vinyl alcohol) hydrogel growth factor release vehicle for wound healing applications. AAPS PharmSci. 2003;5(4):E33. PMID: 15198521; PMCID: PMC2750995. https://doi.org/10.1208/ps050433
- Barrett D.A., Hartshome M.S., Hussain M.A., Shaw P.N., Davies M.C. Resistance to nonspecific protein adsorption by poly(vinyl alcohol) thin films adsorbed to a poly(styrene) support matrix studied using surface plasmon resonance. Anal Chem. 2001;73(21):5232-5239. PMID: 11721924. https://doi.org/10.1021/ac010368u
- Hassan C.M., Peppas N.A. Structure and applications of poly(vinyl alcohol) hydrogels produced by conventional crosslinking or by freezing/thawing methods. Adv Polym Sci. 2000;153:37-65. https://doi.org/10.1007/3-540-46414-X_2
- Dragan E.S. Design and applications of interpenetrating polymer network hydrogels. A review. Chem Eng J. 2014;243:572-590. https://doi.org/10.1016/j.cej.2014.01.065
- Pohan G., Chevallier P., Anderson D.E.J., Tse J.W., Yao Y., Hagen M.W., Mantovani D., Hinds M.T., Yim E.K.F. Luminal plasma treatment for small diameter polyvinyl alcohol tubular scaffolds. Front Bioeng Biotechnol. 2019;7:117. PMID: 31192200; PMCID: PMC6541113. https://doi.org/10.3389/fbioe.2019.00117
- Wan W., Dawn Bannerman A., Yang L., Mak H. Poly(vinyl alcohol) cryogels for biomedical applications. Adv Polym Sci. 2014;263:283-321. https://doi.org/10.1007/978-3-319-05846-7_8
- Chaouat M., Le Visage C., Baille W.E., Escoubet B., Chaubet F., Mateescu M.A., Letourneur D. A novel cross-linked Poly(vinyl alcohol) (PVA) for vascular grafts. Adv Funct Mater. 2008;18(19):2855-2861. https://doi.org/10.1002/adfm.200701261
- Cutiongco M.F.A., Kukumberg M., Peneyra J.L., Yeo M.S., Yao J.Y., Rufaihah A.J., Le Visage C., Ho J.P., Yim E.K.F. Submillimeter diameter poly(vinyl alcohol) vascular graft patency in rabbit model. Front Bioeng Biotechnol. 2016;4:44. PMID: 27376059; PMCID: PMC4896917. https://doi.org/10.3389/fbioe.2016.00044
- Gupta S., Greeshma T., Basu B., Goswami S., Sinha A. Stiffness- and wettability-dependent myoblast cell compatibility of transparent poly(vinyl alcohol) hydrogels. J Biomed Mater Res B Appl Biomater. 2013;101(2):346-354. PMID: 23165741. https://doi.org/10.1002/jbm.b.32845
- Матвеева В.Г., Ханова М.Ю., Глушкова Т.В., Антонова Л.В. Влияние различных концентраций фибриногена на свойства фибриновой матрицы для тканевой сосудистой инженерии. Российский медико-биологический вестник имени академика И.П. Павлова. 2021;29(1):21-34. https://doi.org/10.23888/PAVLOVJ202129121-34 Matveeva V.G., Khanova M.Yu., Glushkova T.V., Antonova L.V. Influence of different concentrations of fibrinogen on the properties of a fibrin matrix for vascular tissue engineering. I.P. Pavlov Russian Medical Biological Herald. 2021;29(1):21-34. (In Russ.) https://doi.org/10.23888/PAVLOVJ202129121-34
- Brown E.E., Zhang J., Laborie M.-P.G. Never-dried bacterial cellulose/fibrin composites: preparation, morphology and mechanical properties. Cellulose. 2011;18(3):631-641. https://doi.org/10.1007/s10570-011-9500-8
- Blat A., Dybas J., Chrabaszcz K., Bulat K., Jasztal A., Kaczmarska M., Pulyk R., Popiela T., Slowik A., Malek K., Adamski M.G., Marzec K.M. FTIR, Raman and AFM characterization of the clinically valid biochemical parameters of the thrombi in acute ischemic stroke. Sci Rep. 2019;9(1):15475. PMID: 31664105; PMCID: PMC6820737. https://doi.org/10.1038/s41598-019-51932-0
- Sa’adon S., Ansari M.N.M., Razak S.I.A., Anand J.S., Nayan N.H.M., Ismail A.E., Khan M.U.A., Haider A. Preparation and physicochemical characterization of a diclofenac sodium-dual layer polyvinyl alcohol patch. Polymers (Basel). 2021;13(15):2459. PMID: 34372062; PMCID: PMC8347342. https://doi.org/10.3390/polym13152459
- Choo K., Ching Y.C., Chuah C.H., Julai S., Liou N.-S. Preparation and characterization of polyvinyl alcohol-chitosan composite films reinforced with cellulose nanofiber. Materials (Basel). 2016;9(8):644. PMID: 28773763; PMCID: PMC5509094. https://doi.org/10.3390/ma9080644
- Lee H., Yamaguchi K., Nagaishi T., Murai M., Kim M., Wei K., Zhang K.-Q., Kim I.S. Enhancement of mechanical properties of polymeric nanofibers by controlling crystallization behavior using a simple freezing/thawing process. RSC Advances. 2017;7:43994-44000. https://doi.org/10.1039/C7RA06545K
- Ino J.M., Chevallier P., Letourneur D., Mantovani D., Le Visage C. Plasma functionalization of poly(vinyl alcohol) hydrogel for cell adhesion enhancement. Biomatter. 2013;3(4):e25414. PMID: 23989063; PMCID: PMC3825233. https://doi.org/10.4161/biom.25414
- Chiu C.L., Hecht V., Duong H., Wu B., Tawil B. Permeability of three-dimensional fibrin constructs corresponds to fibrinogen and thrombin concentrations. Biores Open Access. 2012;1(1):34-40. PMID: 23515363; PMCID: PMC3559212. https://doi.org/10.1089/biores.2012.0211
- du Toit J.-P., Pott R.W.M. Transparent polyvinyl-alcohol cryogel as immobilisation matrix for continuous biohydrogen production by phototrophic bacteria. Biotechnol Biofuels. 2020;13:105. PMID: 32536970; PMCID: PMC7285740. https://doi.org/10.1186/s13068-020-01743-7
- Sau S., Pandit S., Kundu S. Crosslinked poly (vinyl alcohol): Structural, optical and mechanical properties. Surfaces and Interfaces. 2021;25:101198. https://doi.org/10.1016/j.surfin.2021.101198

